2
answers
0
watching
1,947
views
28 Sep 2019
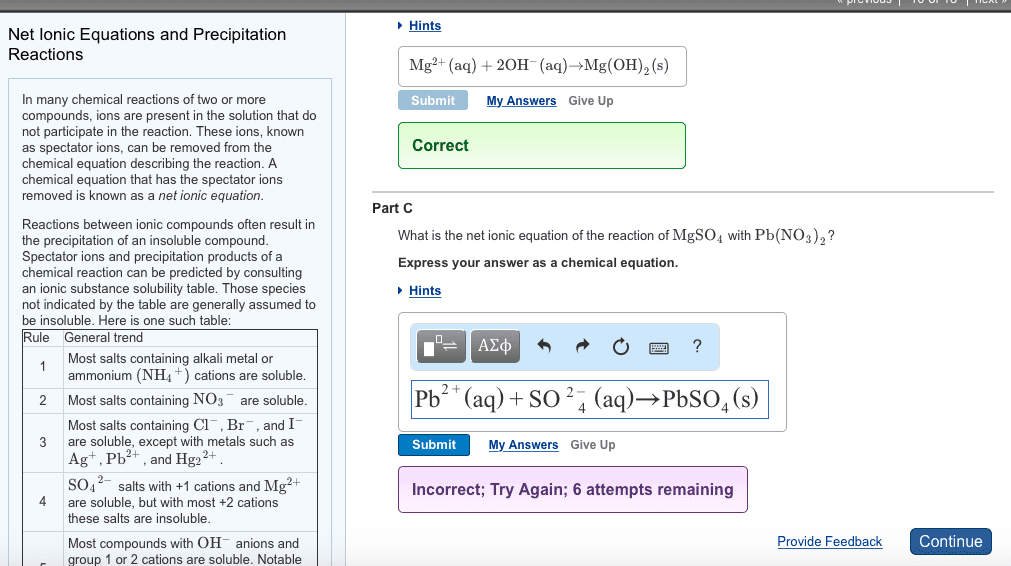
Three solutions are mixed together to form a single solution. One contains 0.2 mol Pb(CH3COO)2 , the second contains 0.1 mol NA2S, and the third contains 0.1 mol CaCl2 . A) Write the net ionic equations for the precipitation reaction or reactions that occur.Express your answer as a balanced net ionic equation. Identify all of the phases in your answer.If there is more than one answer, separate them by a comma.B)What are the spectator ions in the solution?Express your answer as an ion. If there is more than one answer, separate them by a comma.Thanks!
Three solutions are mixed together to form a single solution. One contains 0.2 mol Pb(CH3COO)2 , the second contains 0.1 mol NA2S, and the third contains 0.1 mol CaCl2 . A) Write the net ionic equations for the precipitation reaction or reactions that occur.Express your answer as a balanced net ionic equation. Identify all of the phases in your answer.If there is more than one answer, separate them by a comma.B)What are the spectator ions in the solution?Express your answer as an ion. If there is more than one answer, separate them by a comma.Thanks!
16 Mar 2024
Patrina SchowalterLv2
28 Sep 2019
Already have an account? Log in