2
answers
0
watching
649
views
28 Sep 2019
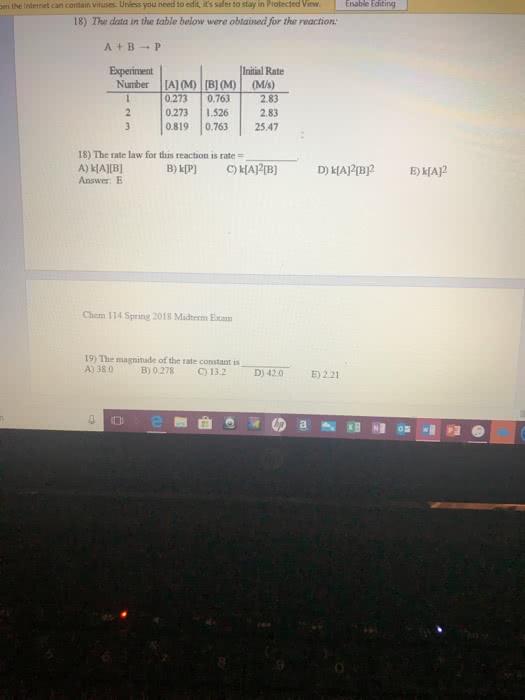
3)The data in the table below were obtained for the reaction: A+ B ----> P Use this info for quest 3 and 4.
3) Write the rate law for this reaction
4) Calculate the magnitude of the rate constant (with units)please include explanation I appreciate it and will rate well thankyou and solved for me so that I may understand for my test please.I get confused on how to get the rate law
Experiment
[A} [M]
[B] [M]
Initial rate
1
0.273
0.763
2.83
2
0.273
1.526
2.83
3
0.819
0.763
24.47
3)The data in the table below were obtained for the reaction: A+ B ----> P Use this info for quest 3 and 4.
3) Write the rate law for this reaction
4) Calculate the magnitude of the rate constant (with units)please include explanation I appreciate it and will rate well thankyou and solved for me so that I may understand for my test please.I get confused on how to get the rate law
Experiment | [A} [M] | [B] [M] | Initial rate |
1 | 0.273 | 0.763 | 2.83 |
2 | 0.273 | 1.526 | 2.83 |
3 | 0.819 | 0.763 | 24.47 |
25 Apr 2023
Collen VonLv2
28 Sep 2019
Already have an account? Log in