2
answers
0
watching
312
views
28 Sep 2019
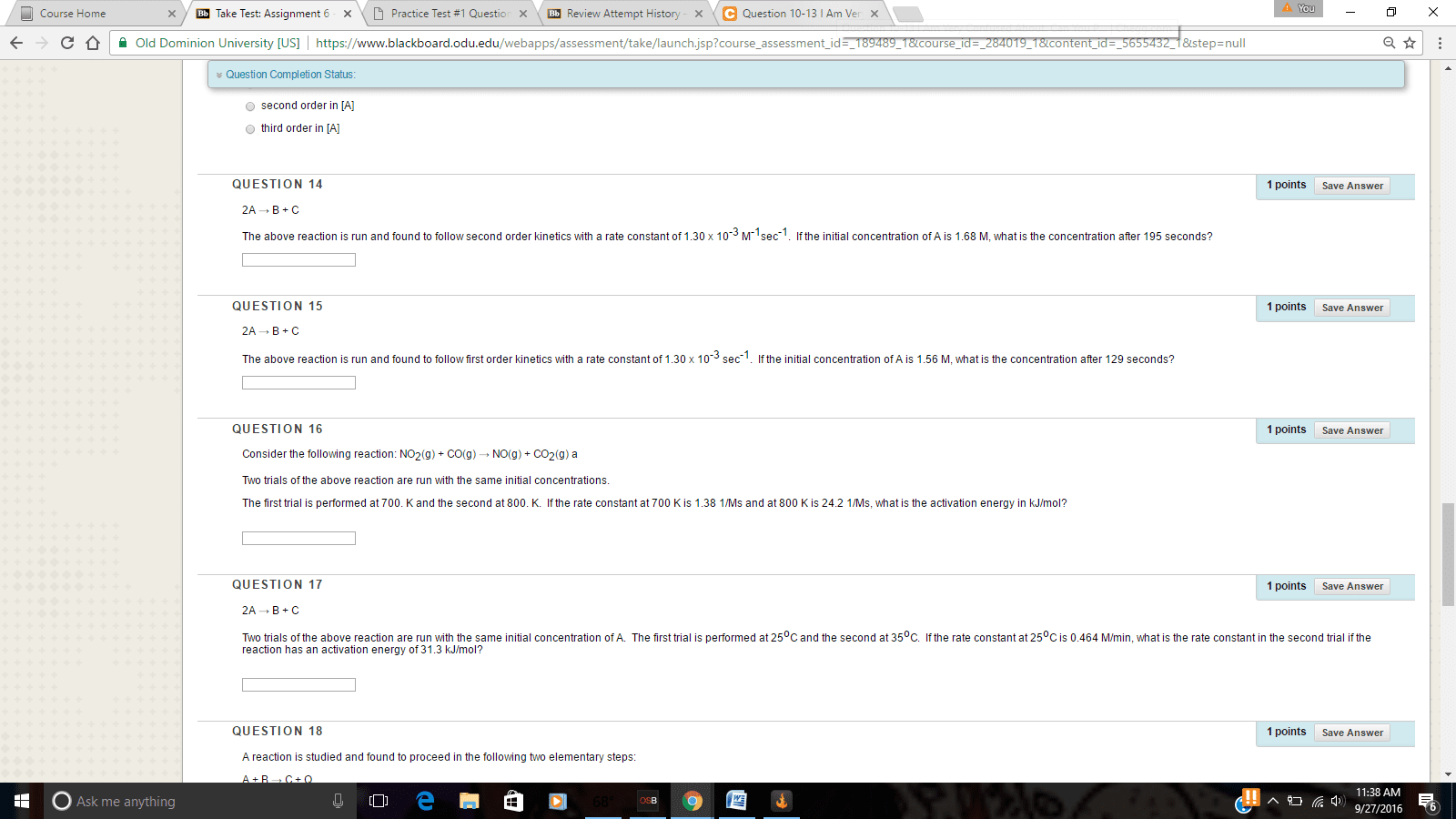
Consider the follwing first order reaction:---------4PH3---> P4 + 6H2----------The half-life of the reaction is 35 seconds at 680 degrees Celcius. Calculate the rate constant for the reaction , and the time required for 95% of the PH3 to decompose.---------8.) The rate constant for the second order reaction below is .54 M^-1 * s^-1. How long in seconds would it take for the concentration of NO2 to decrease from .65 M to .18 M? ------------2NO2---->2NO+O2Show all steps with correct answer for 5 star rating. Thank you.
Consider the follwing first order reaction:---------4PH3---> P4 + 6H2----------The half-life of the reaction is 35 seconds at 680 degrees Celcius. Calculate the rate constant for the reaction , and the time required for 95% of the PH3 to decompose.---------8.) The rate constant for the second order reaction below is .54 M^-1 * s^-1. How long in seconds would it take for the concentration of NO2 to decrease from .65 M to .18 M? ------------2NO2---->2NO+O2Show all steps with correct answer for 5 star rating. Thank you.
2
answers
0
watching
312
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Bunny GreenfelderLv2
28 Sep 2019
Already have an account? Log in
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232