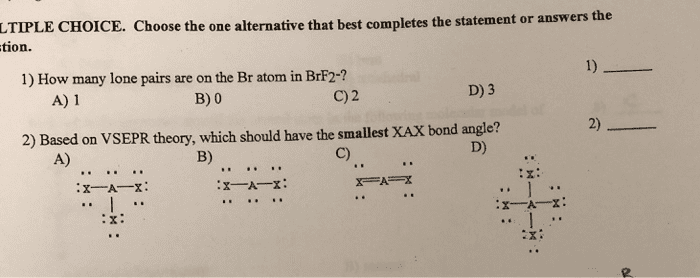1
answer
0
watching
452
views
12 Nov 2019
Answer True or false for the following questions.
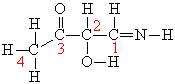
1) There are eleven Ï bonds in this molecule?
2) There are three lone pairs of electrons in the complete Lewisstructure?
3) The C-N bond is formed from overlap of an sp3 hybrid orbitalfrom the carbon atom with an sp2 hybrid orbital from the nitrogenatom?
4) An sp2 hybrid orbital on C-1 overlaps with an sp3 hybrid orbitalon C-2 to form the sigma bond between C-1 and C-2?
5) There are four Ï bonds in this molecule?
6)The C-2 and C-3 atoms cannot rotate about the internuclear axisbetween the two atoms since the Ï bond would break?
7) The Ï bond between C-3 and O is formed by overlap ofunhybridized p atomic orbitals?

Answer True or false for the following questions.
1) There are eleven Ï bonds in this molecule?
2) There are three lone pairs of electrons in the complete Lewisstructure?
3) The C-N bond is formed from overlap of an sp3 hybrid orbitalfrom the carbon atom with an sp2 hybrid orbital from the nitrogenatom?
4) An sp2 hybrid orbital on C-1 overlaps with an sp3 hybrid orbitalon C-2 to form the sigma bond between C-1 and C-2?
5) There are four Ï bonds in this molecule?
6)The C-2 and C-3 atoms cannot rotate about the internuclear axisbetween the two atoms since the Ï bond would break?
7) The Ï bond between C-3 and O is formed by overlap ofunhybridized p atomic orbitals?
1
answer
0
watching
452
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Tod ThielLv2
9 Apr 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232