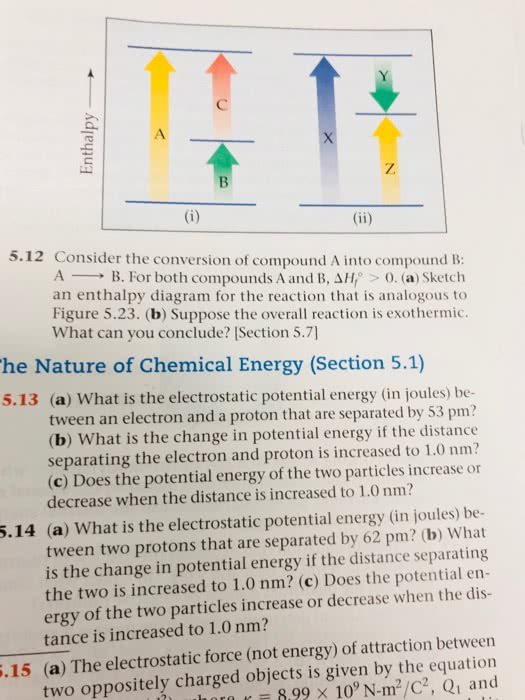
PROBLEM SET 2: INTERACTION ENERGIES AND PROPERTIES OF SOLUTIONS: Information you will need for these problems: Dielectric constant of H2O is 80. Dielectric constant of n-pentane is 1.84. Avogadro constant = 6.022 x 1023 mol-1.Coulomb constant = 9 x 109 Nm2.C2. Charge on an electron = 1.602 x 10-19 C.
1. A chloride ion and a sodium ion are separated by a center-center distance of 0.5 nm. Calculate the interaction energy (the energy required to pull them infinitely apart) if the medium between them is (a) water, or (b) n-pentane. Express the energy in joules per mole of ion pairs.
(c) How far apart would the ions need to be for them to have the same interaction energy as that when they are separated by 0.5 nm in water (i.e. as calculated in part a) if the medium between them is n-pentane? Give your answer in nm.
PROBLEM SET 2: INTERACTION ENERGIES AND PROPERTIES OF SOLUTIONS: Information you will need for these problems: Dielectric constant of H2O is 80. Dielectric constant of n-pentane is 1.84. Avogadro constant = 6.022 x 1023 mol-1.Coulomb constant = 9 x 109 Nm2.C2. Charge on an electron = 1.602 x 10-19 C.
1. A chloride ion and a sodium ion are separated by a center-center distance of 0.5 nm. Calculate the interaction energy (the energy required to pull them infinitely apart) if the medium between them is (a) water, or (b) n-pentane. Express the energy in joules per mole of ion pairs.
(c) How far apart would the ions need to be for them to have the same interaction energy as that when they are separated by 0.5 nm in water (i.e. as calculated in part a) if the medium between them is n-pentane? Give your answer in nm.