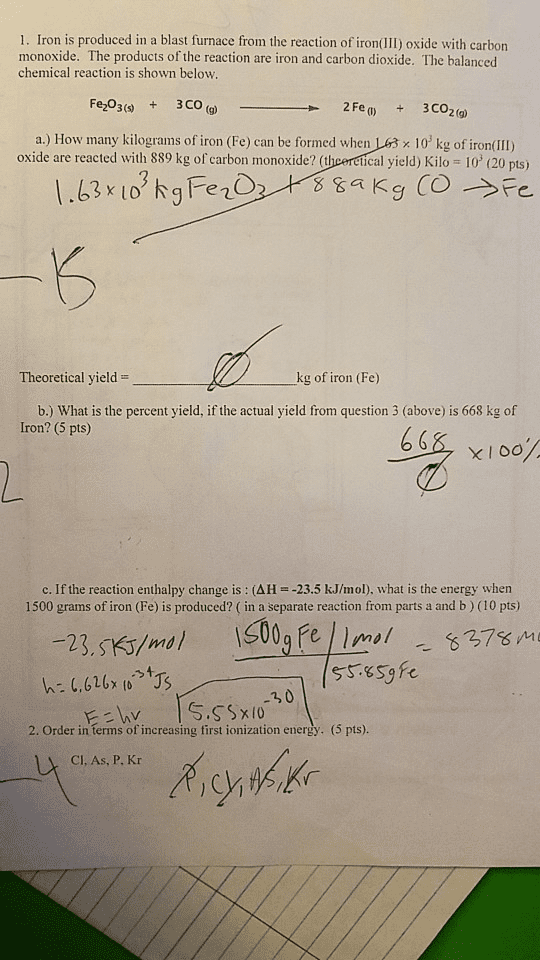1
answer
0
watching
460
views
12 Nov 2019
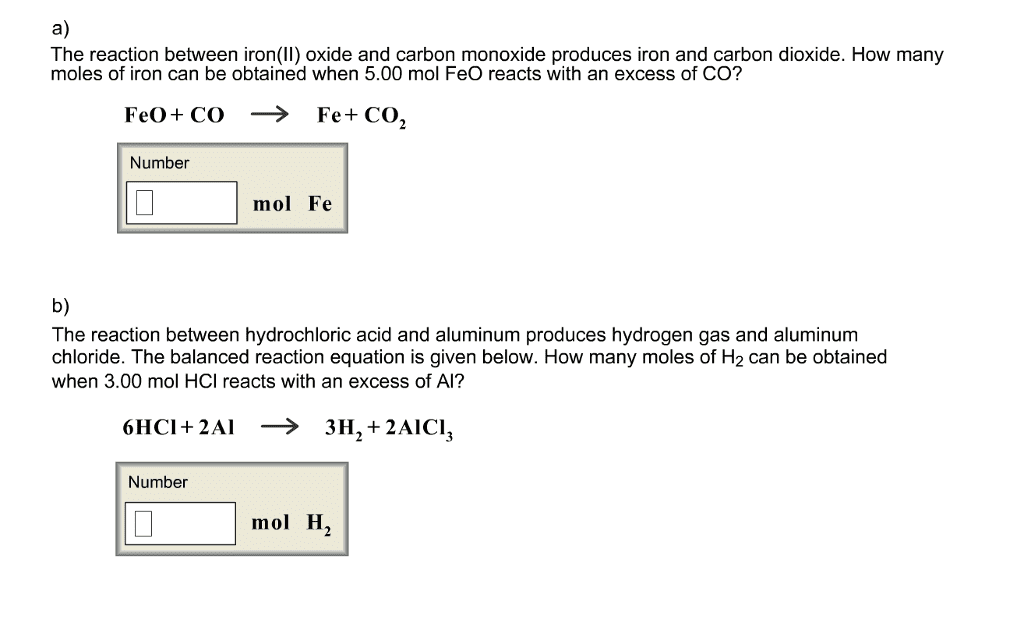
The reaction between iron(II) oxide and carbon monoxide produces iron and carbon dioxide. How many moles of iron can be obtained when 5.75 mol FeO reacts with an excess of CO? FeO+ CO >> Fe+Co2 ___ mol Fe I would prefer a full explanation than the right answer.
The reaction between iron(II) oxide and carbon monoxide produces iron and carbon dioxide. How many moles of iron can be obtained when 5.75 mol FeO reacts with an excess of CO? FeO+ CO >> Fe+Co2 ___ mol Fe I would prefer a full explanation than the right answer.
1
answer
0
watching
460
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Sixta KovacekLv2
28 Jun 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232