1
answer
0
watching
181
views
12 Nov 2019

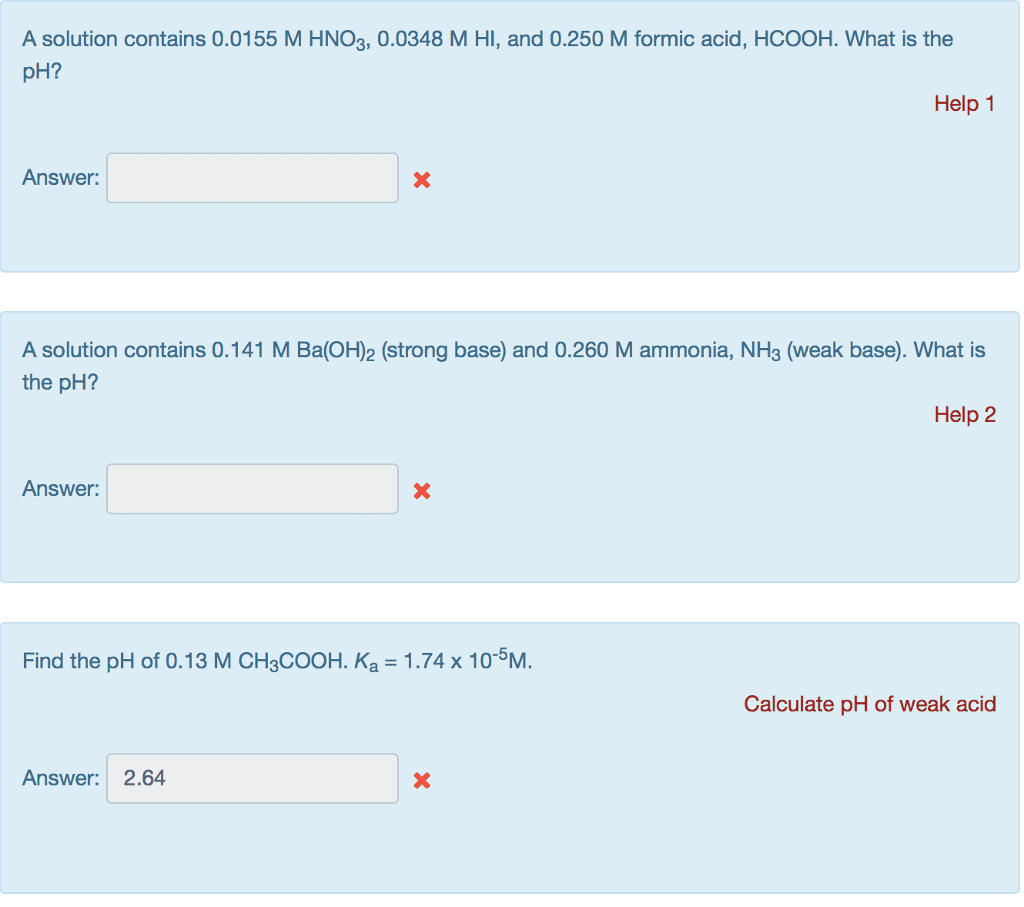
11.5 mL of 0.470 M NaOH is mixed with 33.5 mL of 0.34 M LiOH. What is the molarity of the OH? Str Answer: A solution contains 0.0121 M HBr and 0.240 M acetic acid. What is the pH? Answer: A solution contains 0.0155 M HNO3,0.0348 M HI, and 0.250 M formic acid, HCOOH. What is the pH? Answer: A solution contains 0.141 M Ba(OH)2 (strong base) and 0.260 M ammonia, NH3 (weak base). What is the pH? Answer:

11.5 mL of 0.470 M NaOH is mixed with 33.5 mL of 0.34 M LiOH. What is the molarity of the OH? Str Answer: A solution contains 0.0121 M HBr and 0.240 M acetic acid. What is the pH? Answer: A solution contains 0.0155 M HNO3,0.0348 M HI, and 0.250 M formic acid, HCOOH. What is the pH? Answer: A solution contains 0.141 M Ba(OH)2 (strong base) and 0.260 M ammonia, NH3 (weak base). What is the pH? Answer:
1
answer
0
watching
181
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Trinidad TremblayLv2
16 Sep 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232