How much energy is required to heat 92.1 g acetone (molar mass=58.08 g/mol) from a solid at -154.0°C to a liquid at -42.0°C? The following physical data may be useful.
ÎHfus = 7.27 kJ/mol
Cliq = 2.16 J/g°C
Cgas = 1.29 J/g°C
Csol = 1.65 J/g°C
Tmelting = -95.0°C
How much energy is required to heat 92.1 g acetone (molar mass=58.08 g/mol) from a solid at -154.0°C to a liquid at -42.0°C? The following physical data may be useful.
ÎHfus = 7.27 kJ/mol
Cliq = 2.16 J/g°C
Cgas = 1.29 J/g°C
Csol = 1.65 J/g°C
Tmelting = -95.0°C
For unlimited access to Homework Help, a Homework+ subscription is required.
Related textbook solutions
Basic Chemistry
Principles of Chemistry Molecular Approach
Chemistry: Structure and Properties
Principles of Chemistry Molecular Approach
Chemistry: A Molecular Approach
Chemistry: A Molecular Approach
Principles of Chemistry: A Molecular Approach
Chemistry: The Central Science
Related questions
1) - A solution was prepared by dissolving 43.0 g of KCl in 225 g of water.
Part A
Calculate the mass percent of KCl in the solution.
Express your answer with the appropriate units.
| | ||
Part B
Calculate the mole fraction of the ionic species KCl in the solution.
Express the concentration numerically as a mole fraction in decimal form.
| | |||
| X = | ? |
Part C
Calculate the molarity of KCl in the solution if the total volume of the solution is 239 mL.
Express your answer with the appropriate units.
| | ||
Part D
Calculate the molality of KCl in the solution.
Express your answer with the appropriate units.
2)- How much energy is required to heat 90.7 g acetone (molar mass=58.08 g/mol) from a solid at -154.0°C to a liquid at -42.0°C? The following physical data may be useful.
ÎHfus = 7.27 kJ/mol
Cliq = 2.16 J/g°C
Cgas = 1.29 J/g°C
Csol = 1.65 J/g°C
Tmelting = -95.0°C
3)- The decomposition of XY is second order in XY and has a rate constant of 7.12Ã10â3 Mâ1â sâ1 at a certain temperature.
Part A
How long will it take for the concentration of XY to decrease to 12.5% of its initial concentration when the initial concentration is 0.200 M?
Part B
If the initial concentration of XY is 0.050 M, what is the concentration of XY after 600 s ?
4) - This table displays the vapor pressure of a substance at several different temperatures. 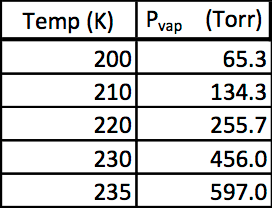
Part A
Use the data to determine the heat of vaporization of the substance in units of kJ/mol. Hint: use Excel
