0
answers
0
watching
547
views
12 Nov 2019


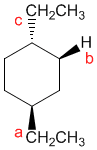
ball & sticklabels ame the above compound, identify each substitutent as axial or equatorial, and tell whether the conformation shown is the more stable or less stable form Omit customary italics from your name Substitutents RI and R2 are designated in the order they appear in the IUPAC name Axial equatorial strain energy increments are available in the table below 1 The IUPAC name ss 2Position of R 3 The conformation shown is the Position of R2 d Cyclohexanesab
0
answers
0
watching
547
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232
Related questions

Name the above compound, identify each substitutent as axial or equatorial, and tell whether the conformation shown is the more stable or less stable form.
Omit customary italics from your name.
Substitutents R1 and R2 are designated in the order they appear in the IUPAC name. Axial/equatorial strain energy increments are available in the table below.
The IUPAC name is
Position of R1 _________axial or equatorial. Position of R2 _________axial or equatorial
The conformation shown is the _________more stable or less stable.
| Substituent (solvent) | kJ/mol | kcal/mol | Substituent (solvent) | kJ/mol | kcal/mol |
|---|---|---|---|---|---|
| -CH3 | 7.3 | 1.7 | -CO2CH2CH3 | 5.0 | 1.2 |
| -CH2CH3 | 7.5 | 1.8 | -Cl | 2.5 | 0.6 |
| -CH(CH3)2 | 9.2 | 2.2 | -Br | 2.5 | 0.6 |
| -C(CH3)3 | 20 | 4.8 | -CN, cyano | 0.8 | 0.2 |
| phenyl | 11.7 | 2.8 | -OH (cyclohexane) | 2.5 | 0.6 |
| cyclohexyl | 9.2 | 2.2 | -OH (isopropanol) | 4.0 | 1.0 |
| -CH=CH2 | 6.2 | 1.5 | -OCH3 | 2.5 | 0.6 |
| -CCH, ethynyl | 2.1 | 0.5 | -NH2 (toluene) | 5.2 | 1.2 |
| -CHO | 3.0 | 0.7 | -NH2 (CH3OCH2CH2OH/H2O) | 7.1 | 1.7 |
| -COCH3 | 5.1 | 1.2 | -NO2 | 4.8 | 1.1 |
| -CO2H | 5.9 | 1.4 |
Energy difference between axial and equatorial conformers.
Solvent specified when the increment is solvent dependent.