1
answer
0
watching
429
views
12 Nov 2019
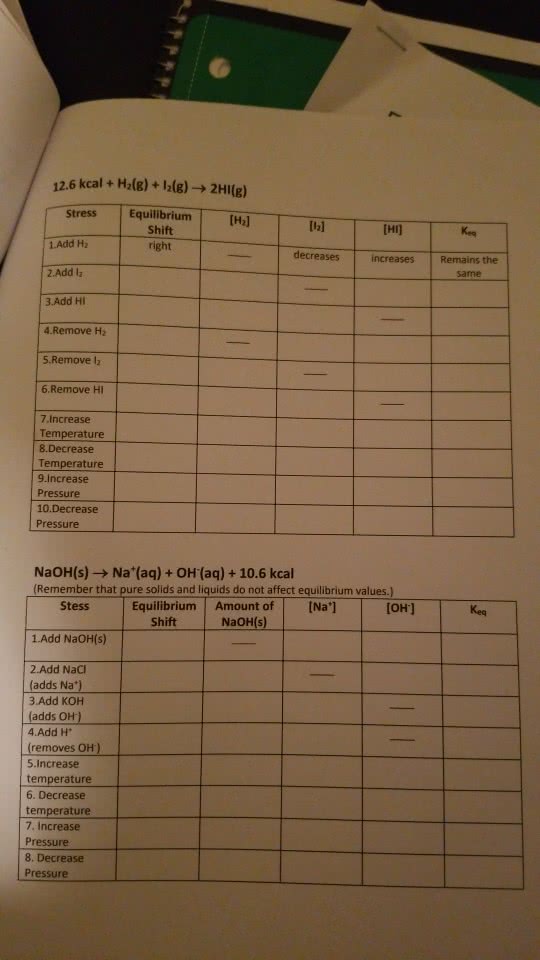
12.6 kcal + Hz(g) +12(g) â 2HI(g) Stress Equilibrium [H2] Shift right 1.Add H2 2.Add l2 3.Add HI 4 Remove H2 5.Remove l2 6.Remove HI decreasesi increases Remains the same 7.Increase 8 Decrease ure 9.Increase Pressure 10.Decrease Pressure NaOH(s) â Na+(aq) + OH(aq) + 10.6 kcal (Remember that pure solids and liquids do not affect equilibrium values.) Stess Equilibrium Amount of[Na] [OH] Shift NaOH(s) 1.Add NaOH(S) 2.Add NaCI 3.Add KOH (adds Na") adds OH 4.Add H 5.Increase temperature 6. Decrease temperature 7. Increase Pressure 8. Decrease Pressure

12.6 kcal + Hz(g) +12(g) â 2HI(g) Stress Equilibrium [H2] Shift right 1.Add H2 2.Add l2 3.Add HI 4 Remove H2 5.Remove l2 6.Remove HI decreasesi increases Remains the same 7.Increase 8 Decrease ure 9.Increase Pressure 10.Decrease Pressure NaOH(s) â Na+(aq) + OH(aq) + 10.6 kcal (Remember that pure solids and liquids do not affect equilibrium values.) Stess Equilibrium Amount of[Na] [OH] Shift NaOH(s) 1.Add NaOH(S) 2.Add NaCI 3.Add KOH (adds Na") adds OH 4.Add H 5.Increase temperature 6. Decrease temperature 7. Increase Pressure 8. Decrease Pressure
1
answer
0
watching
429
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Nestor RutherfordLv2
19 Mar 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232


