1
answer
0
watching
1,056
views
11 Nov 2019
The following figure represents solutions that occur at various stages during the titration of a weak acid, HA, with NaOH. (The Na+ ions and water have been omitted for clarity.) Which of the following statements are true?
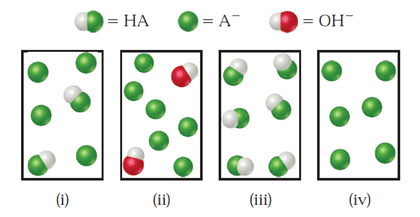
(i) The equivalence point is represented in (iv).
(ii) Both (i) and (ii) are buffer solutions.
(iii) The highest pH is represented by (ii).
A) statements (i) and (ii) are true.
B) only one statement is true.
C) all three statements are true.
D) statements (ii) and (iii) are true.
E) statements (i) and (iii) are true.
(iv)
The following figure represents solutions that occur at various stages during the titration of a weak acid, HA, with NaOH. (The Na+ ions and water have been omitted for clarity.) Which of the following statements are true?

(i) The equivalence point is represented in (iv).
(ii) Both (i) and (ii) are buffer solutions.
(iii) The highest pH is represented by (ii).
A) statements (i) and (ii) are true.
B) only one statement is true.
C) all three statements are true.
D) statements (ii) and (iii) are true.
E) statements (i) and (iii) are true.
(iv)
Casey DurganLv2
27 Sep 2019
