1
answer
0
watching
1,892
views
11 Nov 2019
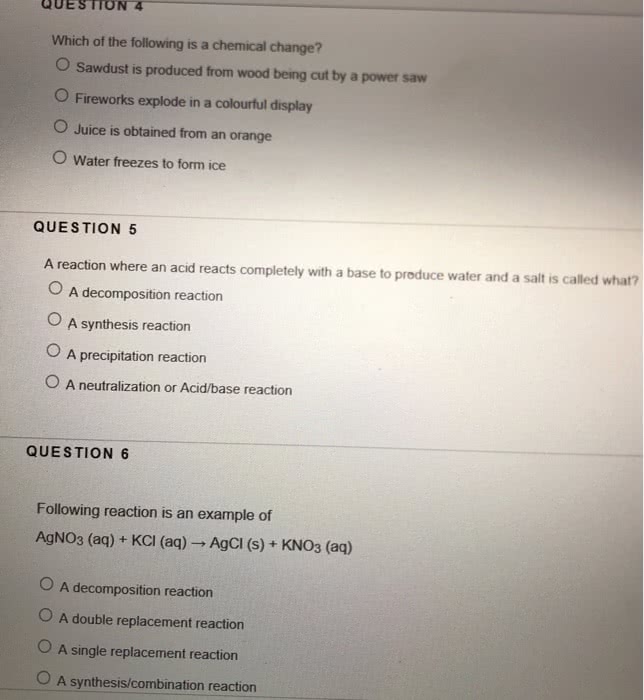
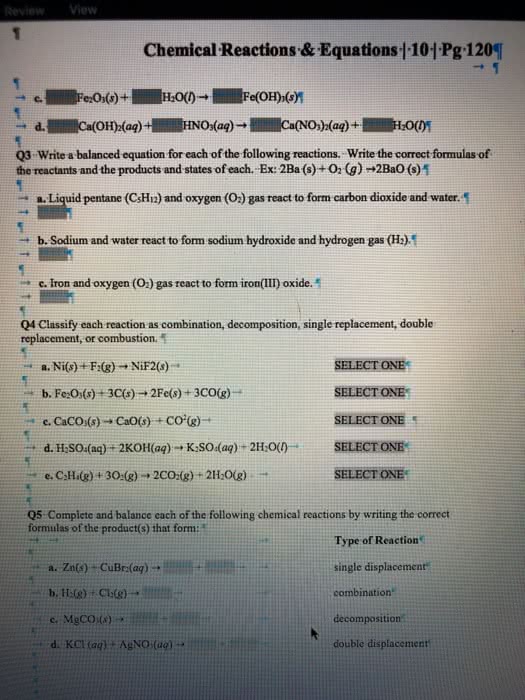
Classify each chemical reaction:
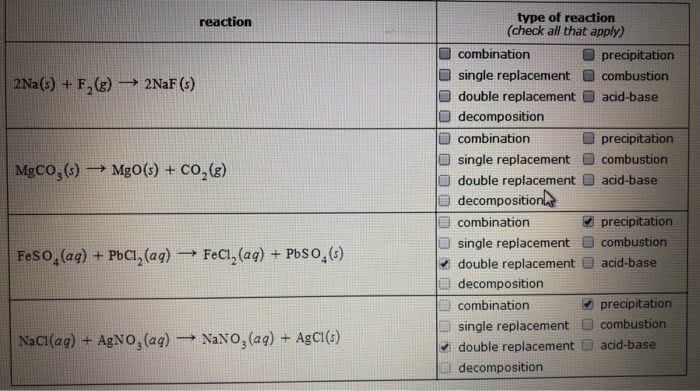
Classify each chemical reaction:

type of reaction (check all that apply) reaction â precipitation combination single replacement combustion double replacement acid-base decomposition combination 2Na(s) + F2(g) â 2Nar(s) precipitation â single replacement combustion â double replacement a acid-base â decompositio MgCO3(s) â MgO(s) + CO2(g) nl precipitation combination single replacement combustion double replacement acid-base decomposition combination single replacement combustion double replacement acid-base decomposition Fes o4(ag) + PbCl2(aq) â FeCl2(aq) Pbso4(s) precipitation Naci(ag) + AgNO3(ag) â NaNO3 (aq) + Agci(s)
Beverley SmithLv2
19 Apr 2019