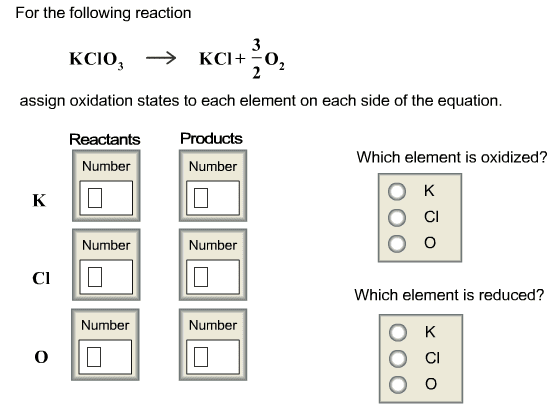
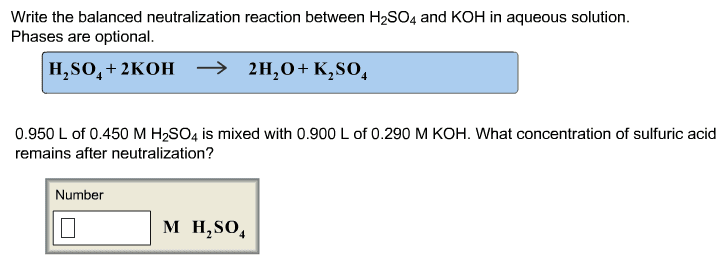
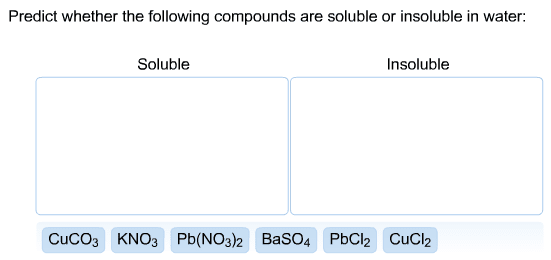
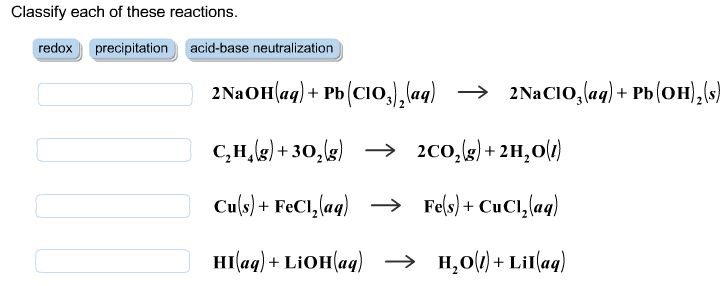Write the balanced complete ionic equations and net ionic equations for the reactions that occur when each of the following solutions are mixed. (Type your answers using the format [NH4]+ for NH4+ or Ca3(PO4)2 for Ca3(PO4)2. Use the lowest possible coefficients.) (a) Fe2(SO4)3(aq) and CaCl2(aq) complete ionic equation: (aq) + SO42-(aq) + Ca2+(aq) + Cl-(aq) (s) + Fe3+(aq) + (aq) net ionic equation: Ca2+(aq) + (aq) (s) (b) FeCl3(aq) and Ag2SO4(aq) complete ionic equation: (aq) + Cl-(aq) + Ag+(aq) + SO42-(aq) (s) + Fe3+(aq) + (aq) net ionic equation: Ag+(aq) + (aq) (s) (c) Na2CO3(aq) and MgSO4(aq) complete ionic equation: (aq) + CO32-(aq) + Mg2+(aq) + SO42-(aq) (s) + Na+(aq) + (aq) net ionic equation: Mg2+(aq) + (aq) (s)
Write the balanced complete ionic equations and net ionic equations for the reactions that occur when each of the following solutions are mixed. (Type your answers using the format [NH4]+ for NH4+ or Ca3(PO4)2 for Ca3(PO4)2. Use the lowest possible coefficients.) (a) Fe2(SO4)3(aq) and CaCl2(aq) complete ionic equation: (aq) + SO42-(aq) + Ca2+(aq) + Cl-(aq) (s) + Fe3+(aq) + (aq) net ionic equation: Ca2+(aq) + (aq) (s) (b) FeCl3(aq) and Ag2SO4(aq) complete ionic equation: (aq) + Cl-(aq) + Ag+(aq) + SO42-(aq) (s) + Fe3+(aq) + (aq) net ionic equation: Ag+(aq) + (aq) (s) (c) Na2CO3(aq) and MgSO4(aq) complete ionic equation: (aq) + CO32-(aq) + Mg2+(aq) + SO42-(aq) (s) + Na+(aq) + (aq) net ionic equation: Mg2+(aq) + (aq) (s)
For unlimited access to Homework Help, a Homework+ subscription is required.