1
answer
1
watching
1,911
views
11 Nov 2019
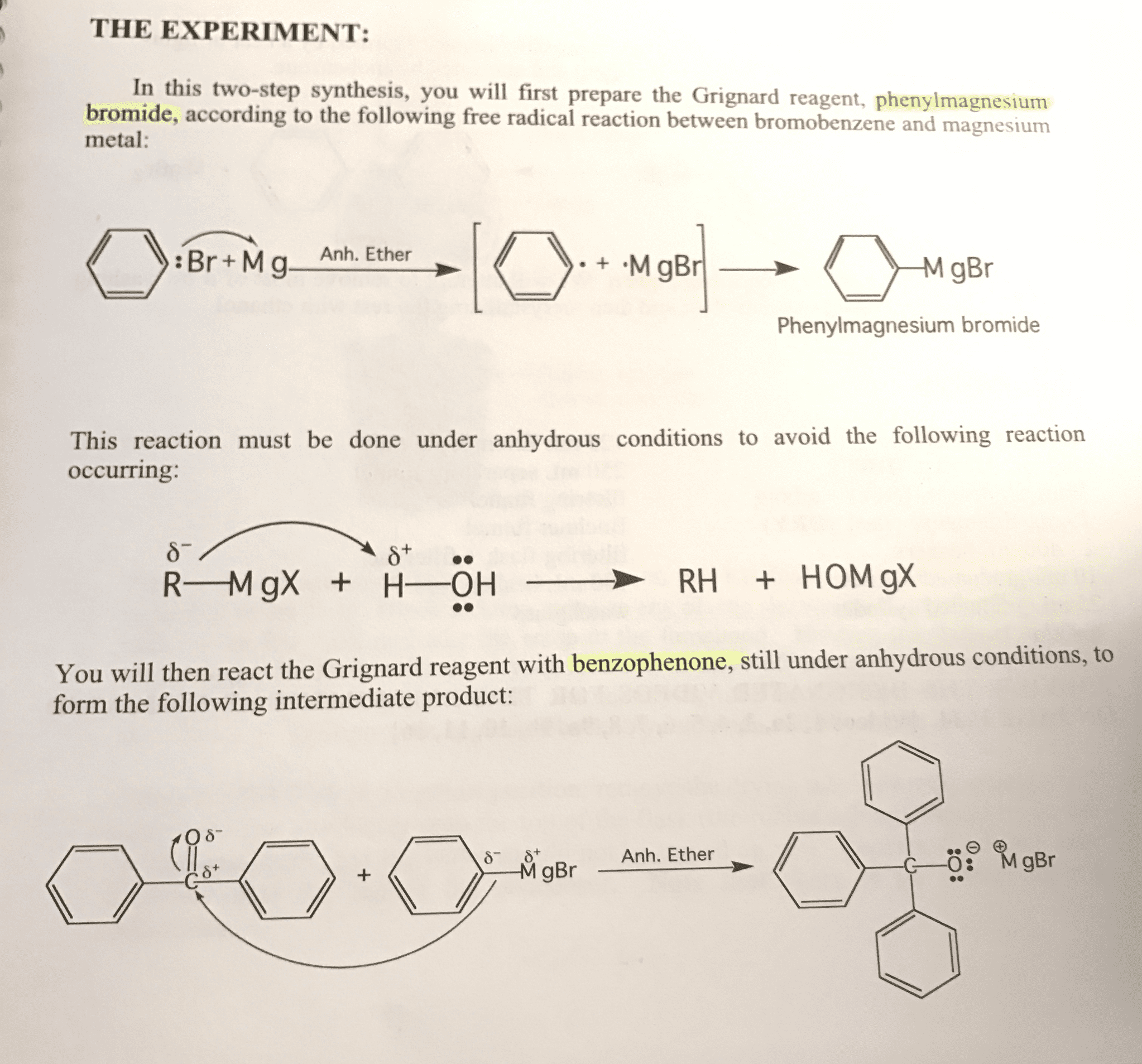
Methyl benzoate was combined with phenylmagnesium bromide to produce triphenylmethanol. This reaction was done in anhydrous conditions. What by-product would be expected if traces of water were present? Write an equation to illustrate the formation of this by-product.
Methyl benzoate was combined with phenylmagnesium bromide to produce triphenylmethanol. This reaction was done in anhydrous conditions. What by-product would be expected if traces of water were present? Write an equation to illustrate the formation of this by-product.
Nelly StrackeLv2
27 Aug 2019