1
answer
0
watching
622
views
11 Nov 2019


Why is it wrong?
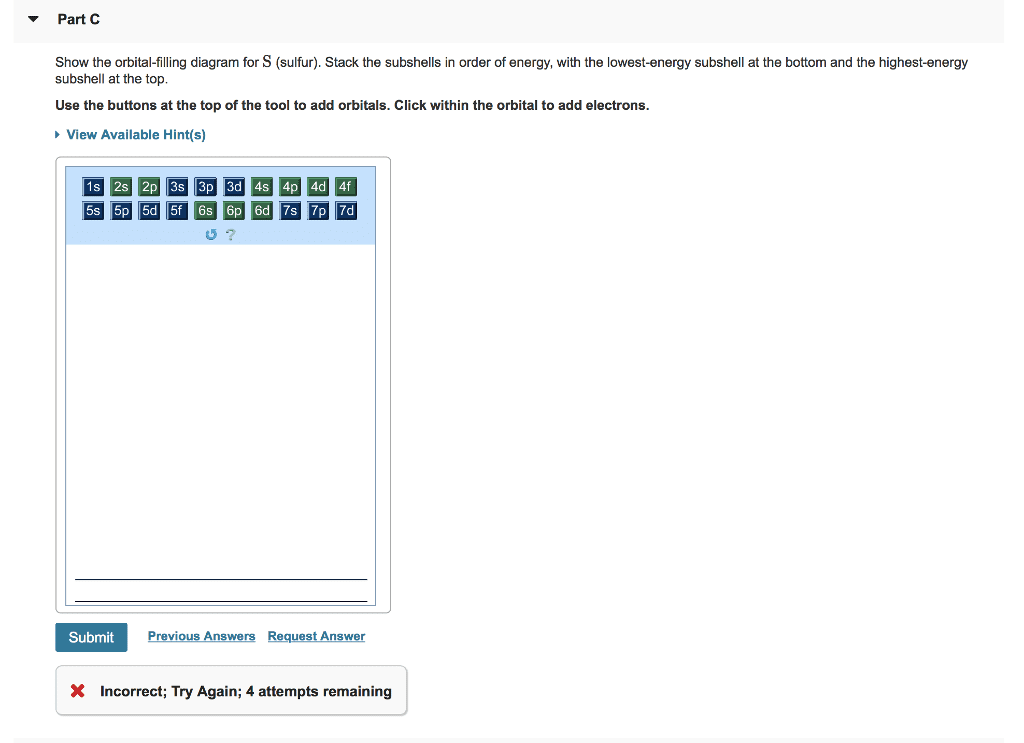
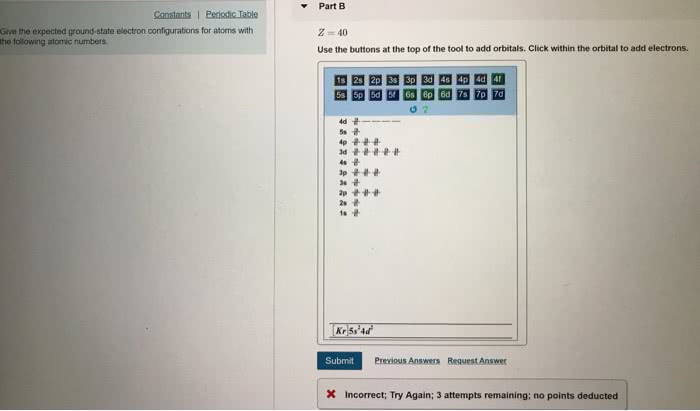
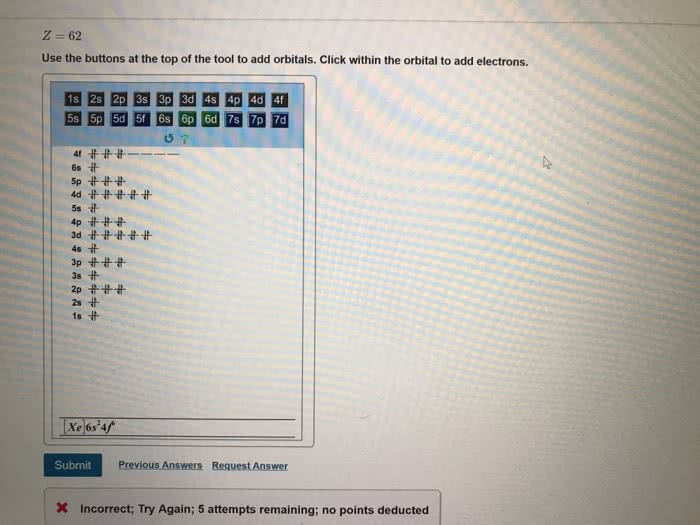
Part B Give the expected ground-state electron configurations for atoms with the following atomic numbers. z=40 Use the buttons at the top of the tool to add orbitals. Click within the orbital to add electrons. 4p 1p ì©ìì 곧. Submit Previous Answers Request Answer X Incorrect; Try Again; 3 attempts remaining: no points deducted
Jarrod RobelLv2
16 May 2019