1
answer
0
watching
600
views
11 Nov 2019
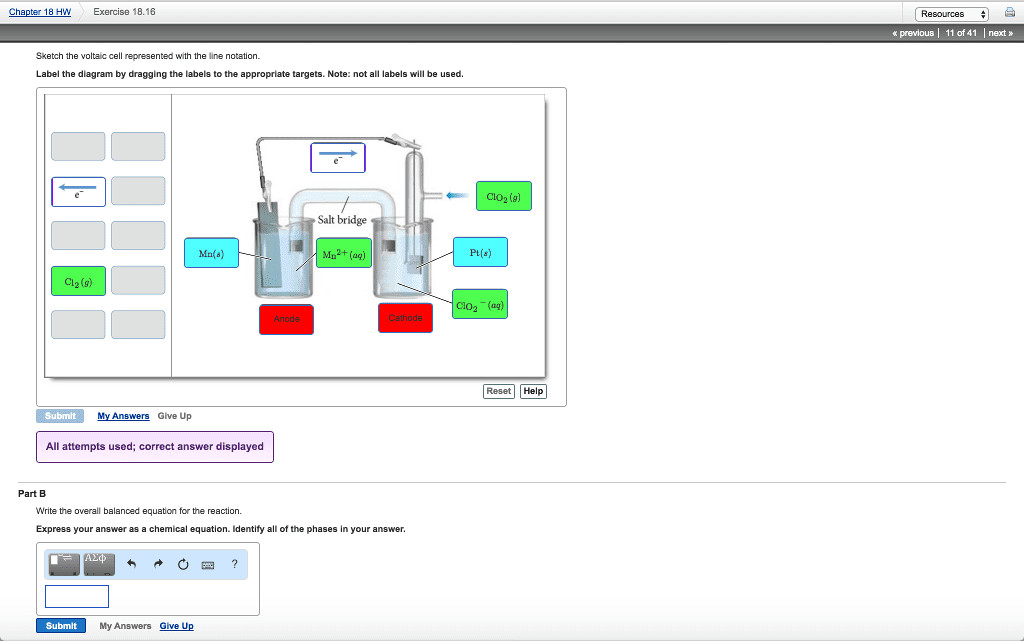
Consider the voltaic cell represented by the following line notation.
Ni(s) | Ni2+(aq) || NO3â(aq),H+(aq),H2O(l) | NO(g) | Pt(s)
Write the overall balanced equation for the reaction.
Express your answer as a chemical equation. Identify all of the phases in your answer.
Consider the voltaic cell represented by the following line notation.
Ni(s) | Ni2+(aq) || NO3â(aq),H+(aq),H2O(l) | NO(g) | Pt(s)
Write the overall balanced equation for the reaction.
Express your answer as a chemical equation. Identify all of the phases in your answer.
Trinidad TremblayLv2
18 Oct 2019