0
answers
0
watching
518
views
11 Nov 2019


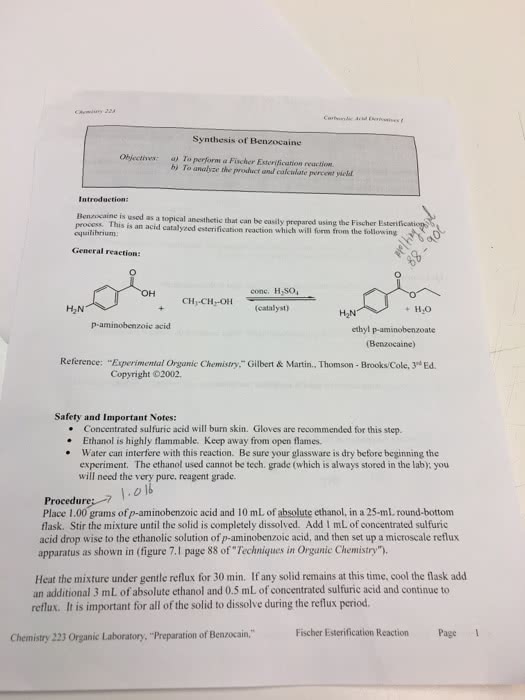
Questions: Turn in with Lab Report I. Why is absolute ethanol rather than 95% ethanol used in this experiment? Why is excess ethanol used for this experiment? What would be the result if one equivalent of ethanol was used? 2. Fischer Esterification Reaction Page Chemistry 223 Organic Laboratory, "Preparation of Benzocaine," 3. Why does scratching the glassware help crystallization occur? 4. Why is sodium carbonate used instead of sodium hydroxide to neutralize the acid after the reaction is complete? (What would happen if sodium hydroxide were used?) âExpertinental Organic Chernistry," Gilbert & Martin. Thomson-Brooks Cole, 3rd Ed. Copyright ©2002. Reference: Modified by B.Wilson, Spring 2016 PIC.COLLAGE