1
answer
0
watching
775
views
11 Nov 2019

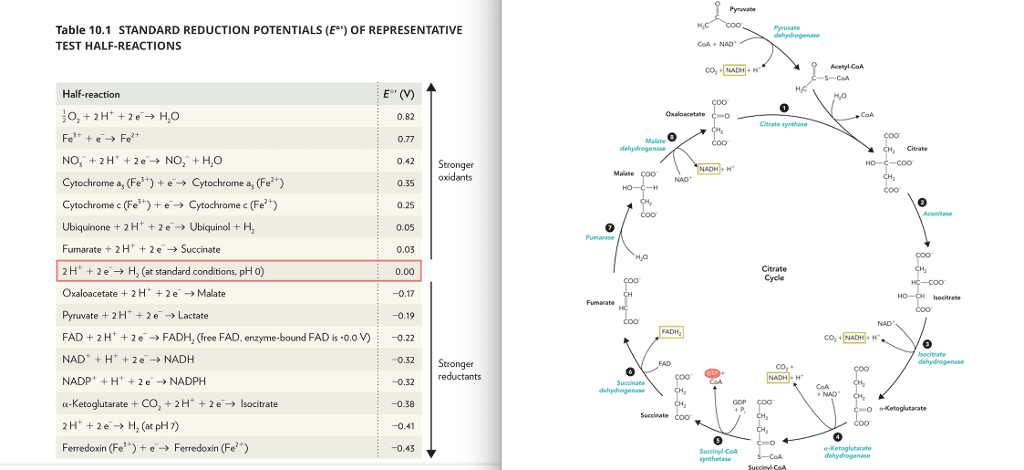
4. Calculate Eã, in Volts and ÎGo, in kJ/mol for the following biochemical redox reactions (all in aqueous solution at pH-7), using the values listed below.) a) CH,CO + H+ + ADH â CH3CH2OH + NAD+ b) 2 Cyt-c[Fe2+1+ NAD+ + H+ â 2 Cyt-Fe3+1+ NADH c)2H2 +02 â 2 H2O d) Pyruvate + NADH â NAD+ + Lactate + f) Succinate + FAD â Fumarate + FADH2 g) L-cystine + H+ + NADH â 2 L-cysteine + NAD Equation Equation CHsCO + 2H+ + 2e-â CH3CH2OH NAD+ + H+ +2e-â NADH -0.20 V 0.32 V 0.25 V 0.18 V -0.395 V 0.22 V 0.03 V 0.34 V ruvate. + 2H' + 2e-â lactate 2H+ + 2e-â H2 O2 + 4H' + 4e-â 2H2ã FAD + 2H+ + 2e-â FADH2 Fumarate-2 + 2H' + 2e-â succinate-2 L-cystine + 2H' + 2e-â 2L-cysteine 1.23 V

4. Calculate Eã, in Volts and ÎGo, in kJ/mol for the following biochemical redox reactions (all in aqueous solution at pH-7), using the values listed below.) a) CH,CO + H+ + ADH â CH3CH2OH + NAD+ b) 2 Cyt-c[Fe2+1+ NAD+ + H+ â 2 Cyt-Fe3+1+ NADH c)2H2 +02 â 2 H2O d) Pyruvate + NADH â NAD+ + Lactate + f) Succinate + FAD â Fumarate + FADH2 g) L-cystine + H+ + NADH â 2 L-cysteine + NAD Equation Equation CHsCO + 2H+ + 2e-â CH3CH2OH NAD+ + H+ +2e-â NADH -0.20 V 0.32 V 0.25 V 0.18 V -0.395 V 0.22 V 0.03 V 0.34 V ruvate. + 2H' + 2e-â lactate 2H+ + 2e-â H2 O2 + 4H' + 4e-â 2H2ã FAD + 2H+ + 2e-â FADH2 Fumarate-2 + 2H' + 2e-â succinate-2 L-cystine + 2H' + 2e-â 2L-cysteine 1.23 V
Nestor RutherfordLv2
28 Jan 2019