1
answer
0
watching
348
views
11 Nov 2019
How many molecules of O2 are needed? (Number 4) and how many formula units of of pottassium chlorate are produced from the reaction of 7.95g potassium chloride? (Last problem)

How many molecules of O2 are needed? (Number 4) and how many formula units of of pottassium chlorate are produced from the reaction of 7.95g potassium chloride? (Last problem)


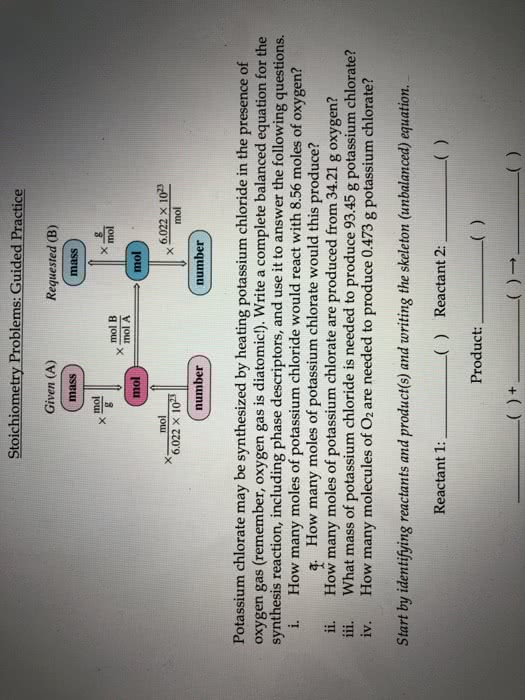
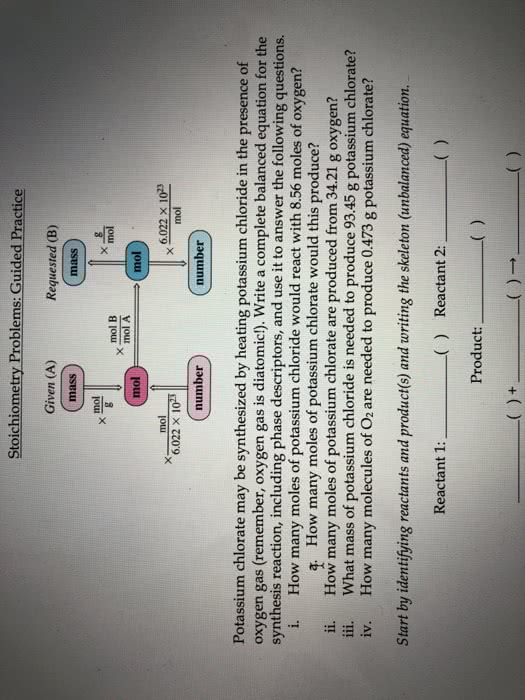
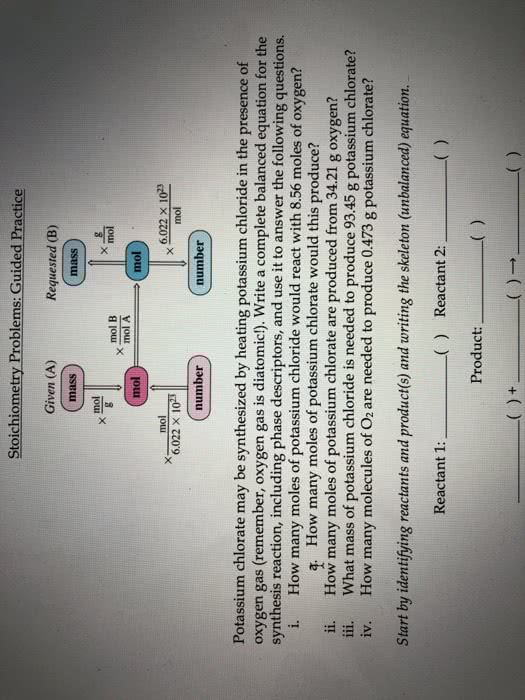
Block: Date: Name: Areté Prep . Chemistry i. How many moles of potassium chloride would react with 8.56 moles of oxygen? mol KCl -mol 02 mol KCl 8.56 mol 02 à How many moles of potassium chlorate would this produce? a. 8.56 mol o2 x mol KCIO i. How many moles of potassium chlorate are produced from 34.21 g oxygen? 1mol 02y mol KCLO 34.21g 02xg02 g 02 x 2 mol KC103 Jn01 02 ii. What mass of potassium chloride is needed to produce 93.45 g potassium chlorate? mol KCl g KCL mol KCIO,1 mol KCI 1 mol KClO3 93.45 g KC103 à g KC iv. How many molecules of O2 are needed to produce 0.473 g potassium chlorate? mol 0 mol KCLO3 -molecules 02 molecules O2 1 mol 02 1 mol KCIO, 0.473 g KCIo3 x .g KC103 = Try this last one yourself How many formula units of potassium chlorate are produced from the reaction of 7.95 g potassium chloride?
Nestor RutherfordLv2
15 Oct 2019