1
answer
2
watching
1,240
views
11 Nov 2019


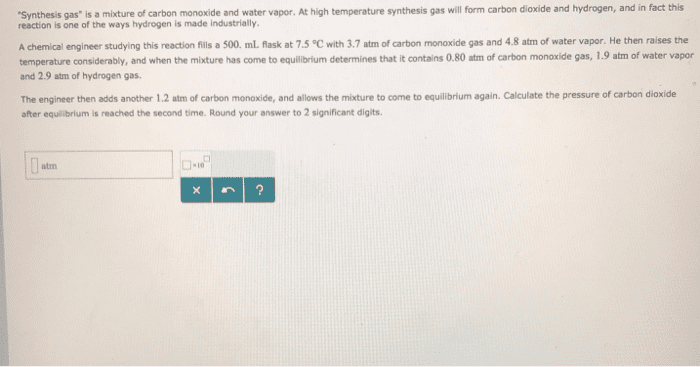
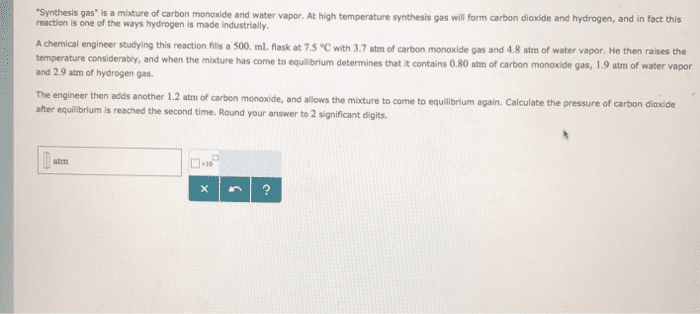
2. The equilibrium constant, Kp, for the reaction CO(g) + H2O(g) CO2(g) + H2(g) at 986 °C is 63. A rigid cylinder at that temperature contains 1.2 atm of carbon monoxide, 1.2 atm of water vapor, 0.30 atm of carbon dioxide and 0.27 atm of hydrogen. (a) Is the system at equilibrium? (b) Which direction must it move to get to equilibrium? e) What are the concentrations of each component at equilibrium?
1
answer
2
watching
1,240
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Liked by mutambonsesheye and 1 others
Keith LeannonLv2
20 Sep 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232