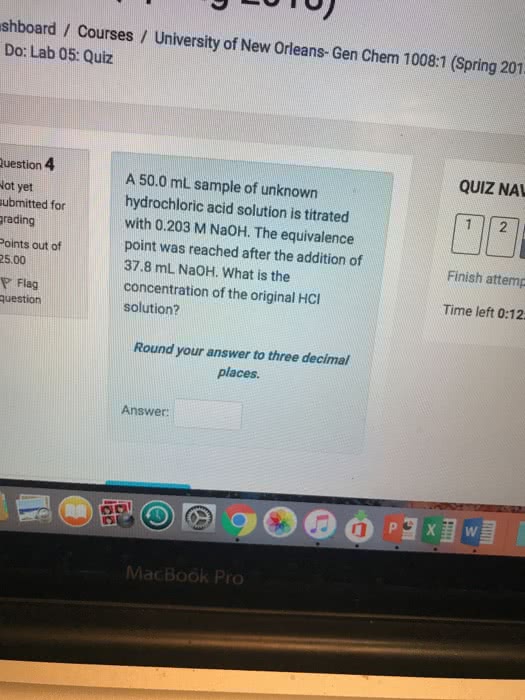
1
answer
0
watching
161
views
11 Nov 2019
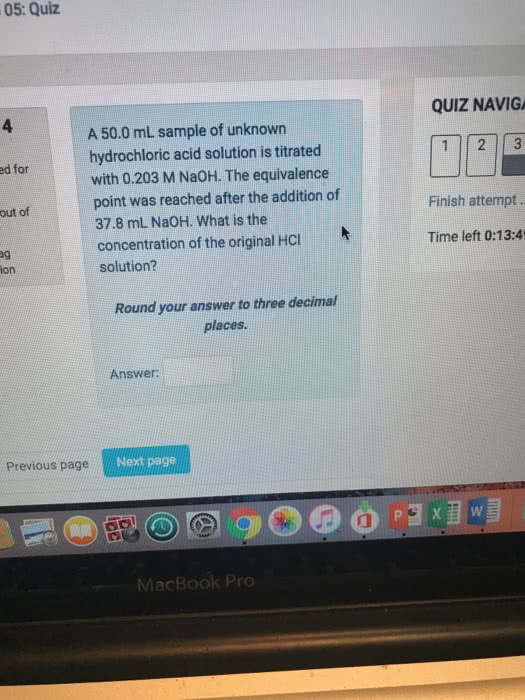

05: Quiz QUIZ NAVIGA 4 A 50.0 mL sample of unknown hydrochloric acid solution is titrated with 0.203 M NaOH. The equivalence point was reached after the addition of 37.8 mL Na0H. What is the concentration of the original HCI solution? ed for out of Finish attempt Time left 0:13:4 ag ion Round your answer to three decimal places Answer Previous page Next page MacBook Pro
1
answer
0
watching
161
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Jamar FerryLv2
26 Apr 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232