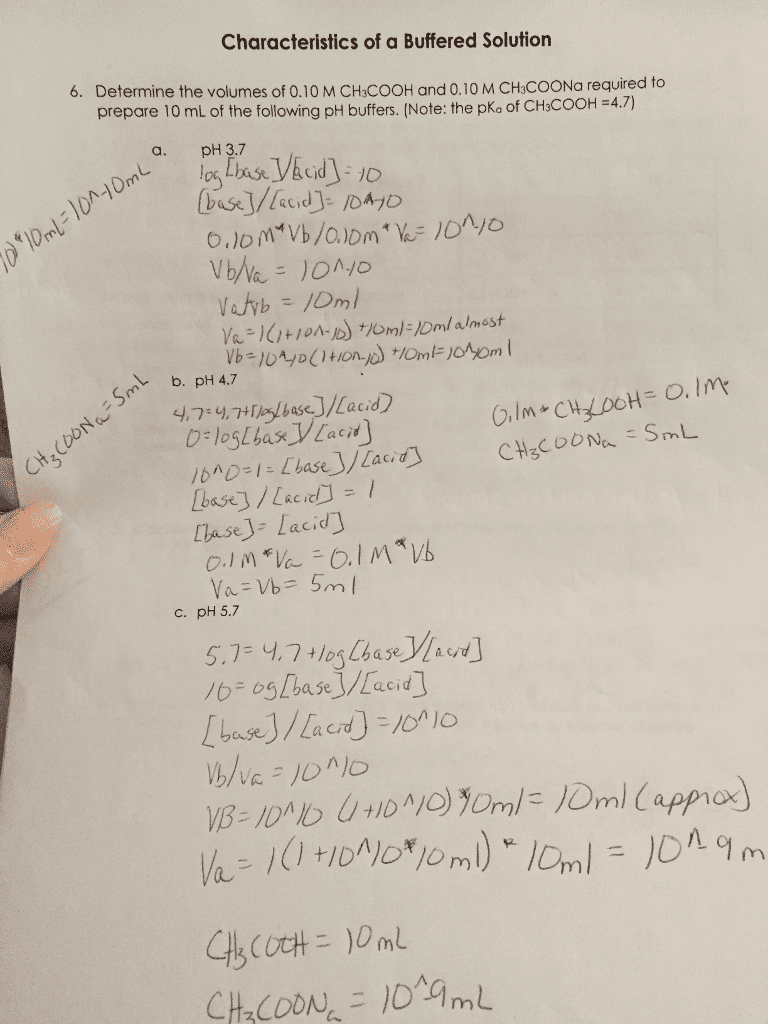1
answer
0
watching
221
views
11 Nov 2019
Calculate the pH of the following buffer solutions: (anwers given in bold)
1) A solution obtained by adding 30 mL of 0.20 M HOAc to 25 mL of 0.10 M NaOH. R = 4.61
2) A solution that is 0.050 M in formic acid and 0.10 M in sodium form. R = 4.0
3) A buffer solution of acetic acid-sodium acetate of pH 5.00 (initial) whose concentration in NaOAc is 0.100 M, after the addition of 10 mL of 0.1 M NaOH to 100 mL of the buffer. R = 5.12
Calculate the pH of the following buffer solutions: (anwers given in bold)
1) A solution obtained by adding 30 mL of 0.20 M HOAc to 25 mL of 0.10 M NaOH. R = 4.61
2) A solution that is 0.050 M in formic acid and 0.10 M in sodium form. R = 4.0
3) A buffer solution of acetic acid-sodium acetate of pH 5.00 (initial) whose concentration in NaOAc is 0.100 M, after the addition of 10 mL of 0.1 M NaOH to 100 mL of the buffer. R = 5.12
Sixta KovacekLv2
22 Sep 2019