1
answer
0
watching
854
views
28 Sep 2019
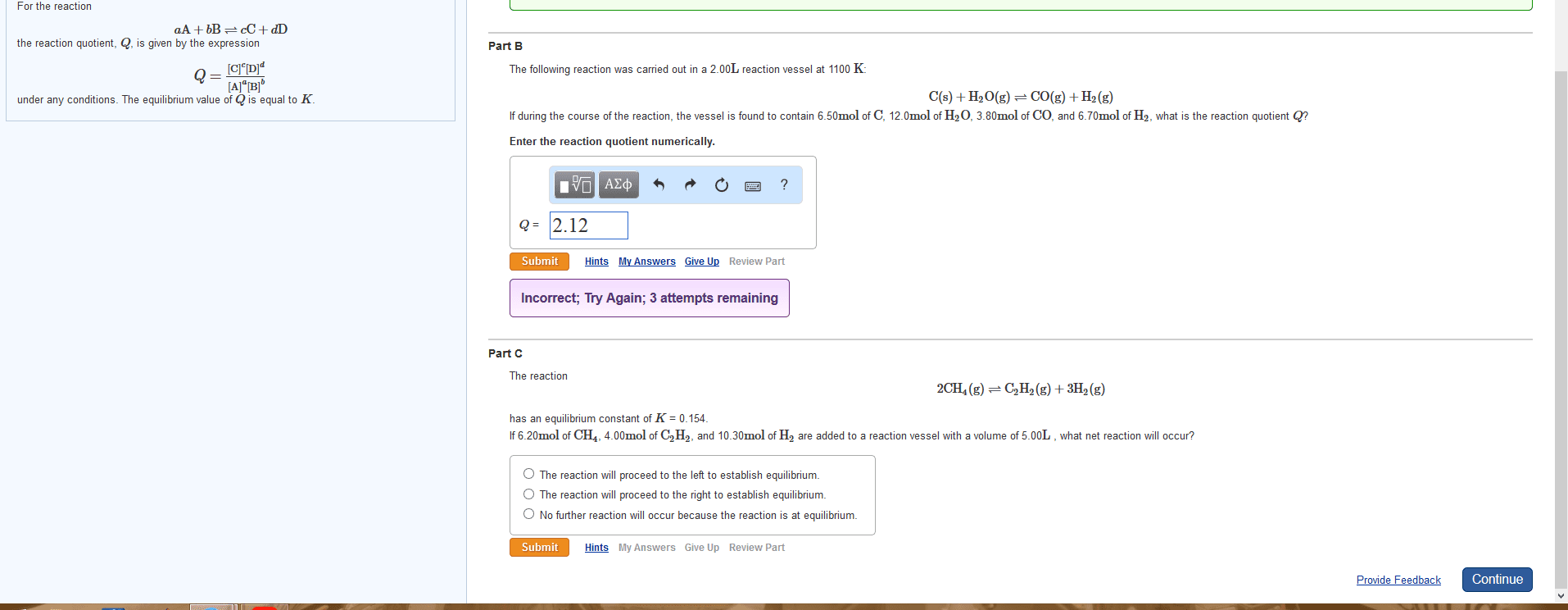
The reaction 2CH4(g)âC2H2(g)+3H2(g) has an equilibrium constant of K = 0.154. If 6.25 mol of CH4, 4.70 mol of C2H2, and 11.30 mol of H2 are added to a reaction vessel with a volume of 5.60 L , what net reaction will occur? The reaction has an equilibrium constant of = 0.154. If 6.25 of , 4.70 of , and 11.30 of are added to a reaction vessel with a volume of 5.60 , what net reaction will occur?
A - The reaction will proceed to the left to establish equilibrium.
B -The reaction will proceed to the right to establish equilibrium.
C -No further reaction will occur because the reaction is at equilibrium.
The reaction 2CH4(g)âC2H2(g)+3H2(g) has an equilibrium constant of K = 0.154. If 6.25 mol of CH4, 4.70 mol of C2H2, and 11.30 mol of H2 are added to a reaction vessel with a volume of 5.60 L , what net reaction will occur? The reaction has an equilibrium constant of = 0.154. If 6.25 of , 4.70 of , and 11.30 of are added to a reaction vessel with a volume of 5.60 , what net reaction will occur?
A - The reaction will proceed to the left to establish equilibrium.
B -The reaction will proceed to the right to establish equilibrium.
C -No further reaction will occur because the reaction is at equilibrium.
Tod ThielLv2
28 Sep 2019