1
answer
0
watching
184
views
11 Nov 2019
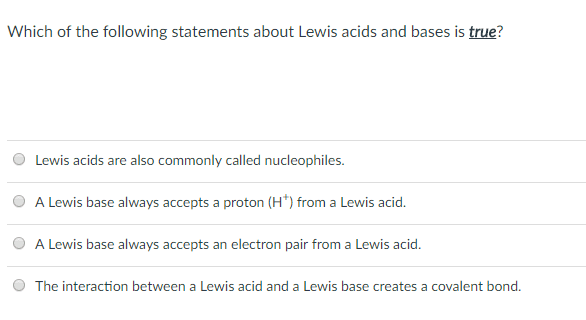

Which of the following statements about Lewis acids and bases is true? Lewis acids are also commonly called nucleophiles. O A Lewis base always accepts a proton (H') from a Lewis acid. O A Lewis base always accepts an electron pair from a Lewis acid The interaction between a Lewis acid and a Lewis base creates a covalent bond.


Which of the following statements about Lewis acids and bases is true? Lewis acids are also commonly called nucleophiles. O A Lewis base always accepts a proton (H') from a Lewis acid. O A Lewis base always accepts an electron pair from a Lewis acid The interaction between a Lewis acid and a Lewis base creates a covalent bond.
1
answer
0
watching
184
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Jean KeelingLv2
27 Oct 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232
Related questions
For the reaction, CO2 + H2O â H2CO3 which of the following statements are incorrect?
| A) | CO2 would be called a Bronsted Lowry Acid and H2O would be called a Bronsted Lowry Base. | |
| B) | H2CO3 forms a coordinate covalent bond by a Lewis acid base reaction. | |
| C) | CO2 would be called a Lewis Acid and H2O would be called a Lewis Base. | |
| D) | The lone pairs in the H2O molecule are used for forma new bond with the carbon in the CO2 molecule. | |
| E) | Both a and d |

