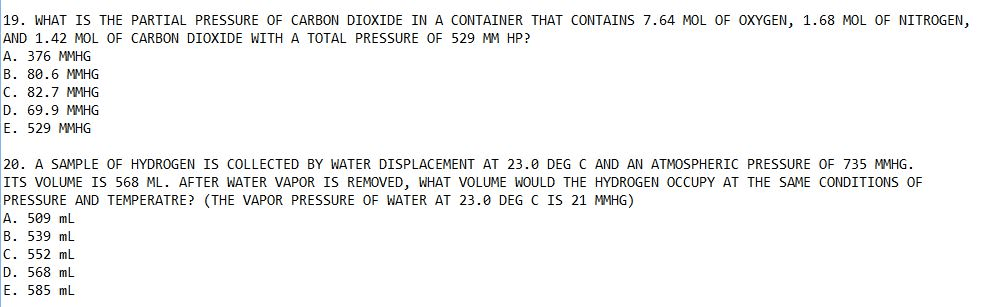
I would like these to be answered thoughtfully and withexplenations for the answers. If I wanted the final answer I wouldlook in te back of the book.
4. Use Hess's Law to find the heat of reaction in the examplebelow.
N2O4 (g) ----> 2NO2 (g)
Given that:
N2O4 (g) ----> N2 (g) + 2O2 (g) (Delta H =-9.7kJ)
2NO2 (g) ----> 2O2(g) + N2(g) ( Delta H= -67.7kJ)
HOW DO WE DECIDE WHICH ONE IS POSITIVE OR NEGATIVE?
8.Calculatethe (Delta H) for this reaction:
SAME AS #4
Zn(s) + Cu(2+ ion)(aq) ---> Zn(2+) + Cu(s)
(Delta H)for Cu(2+) = +64.4 kJ/Mol; (Delta H) for Zn(2+) = -152.4kJ/mol
PLEASEPROVIDE A CLEAR EXPLANATION FOR #17
17. If one mole ofeach of these solutes in added to the same amount of water, whichsolution has the highest boiling point? (glucose, aluminum sulfate,magnesium acetate, copper (I) chloride)
PLEASE PROVIDE CLEAR EXPLANATION
23. With solutions of "strong" acids and "strong" bases, the word"strong" refers to ...
24. What are the products of Al(OH)3 + H2CO3?
FOR #33 and #36 HOW DO WE KNOW THE OX # IF THERE ARE NO OXYGEN ORHYDROGEN?
33. Identify the oxidizing agent in the following reaction: 2Na + S---> Na2S
36. Which atom has a change in oxidation number of +4 in thefollowing reaction?
K2Cr2O7 + H2O + S ----> KOH + Cr2O3 +SO2
I would like these to be answered thoughtfully and withexplenations for the answers. If I wanted the final answer I wouldlook in te back of the book.
4. Use Hess's Law to find the heat of reaction in the examplebelow.
N2O4 (g) ----> 2NO2 (g)
Given that:
N2O4 (g) ----> N2 (g) + 2O2 (g) (Delta H =-9.7kJ)
2NO2 (g) ----> 2O2(g) + N2(g) ( Delta H= -67.7kJ)
8.Calculatethe (Delta H) for this reaction:
Zn(s) + Cu(2+ ion)(aq) ---> Zn(2+) + Cu(s)
(Delta H)for Cu(2+) = +64.4 kJ/Mol; (Delta H) for Zn(2+) = -152.4kJ/mol
PLEASEPROVIDE A CLEAR EXPLANATION FOR #17
17. If one mole ofeach of these solutes in added to the same amount of water, whichsolution has the highest boiling point? (glucose, aluminum sulfate,magnesium acetate, copper (I) chloride)
PLEASE PROVIDE CLEAR EXPLANATION
23. With solutions of "strong" acids and "strong" bases, the word"strong" refers to ...
24. What are the products of Al(OH)3 + H2CO3?
FOR #33 and #36 HOW DO WE KNOW THE OX # IF THERE ARE NO OXYGEN ORHYDROGEN?
33. Identify the oxidizing agent in the following reaction: 2Na + S---> Na2S
36. Which atom has a change in oxidation number of +4 in thefollowing reaction?
K2Cr2O7 + H2O + S ----> KOH + Cr2O3 +SO2
For unlimited access to Homework Help, a Homework+ subscription is required.