1
answer
0
watching
108
views
28 Sep 2019
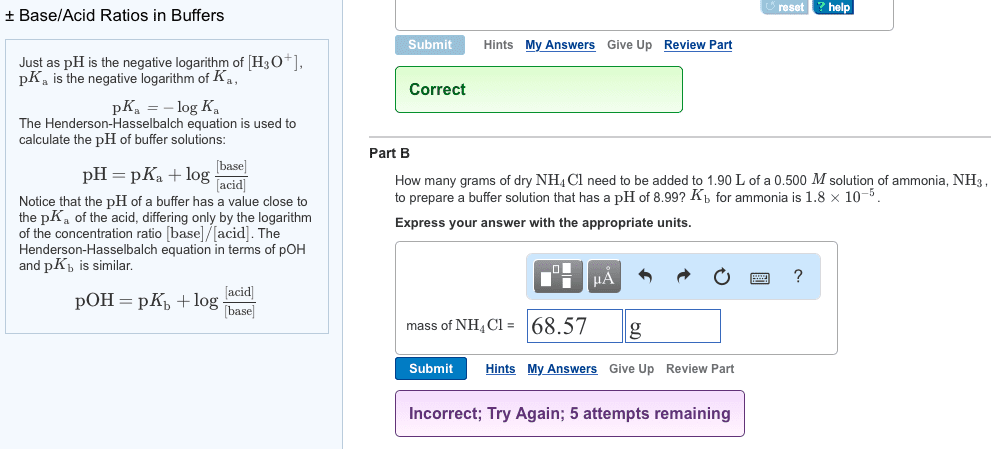
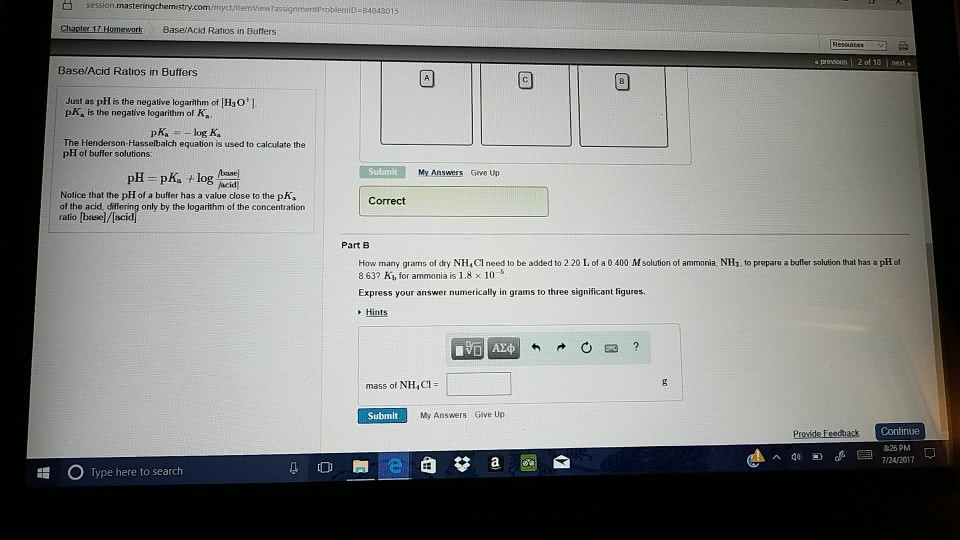
How many grams of dry \rm NH_4Cl need to be added to 2.10 L of a 0.500 \it M solution of ammonia, \rm NH_3, to prepare a buffer solution that has a \rm pH of 8.79? K_b for ammonia is 1.8\times 10^{-5}.
How many grams of dry \rm NH_4Cl need to be added to 2.10 L of a 0.500 \it M solution of ammonia, \rm NH_3, to prepare a buffer solution that has a \rm pH of 8.79? K_b for ammonia is 1.8\times 10^{-5}.
Jarrod RobelLv2
28 Sep 2019