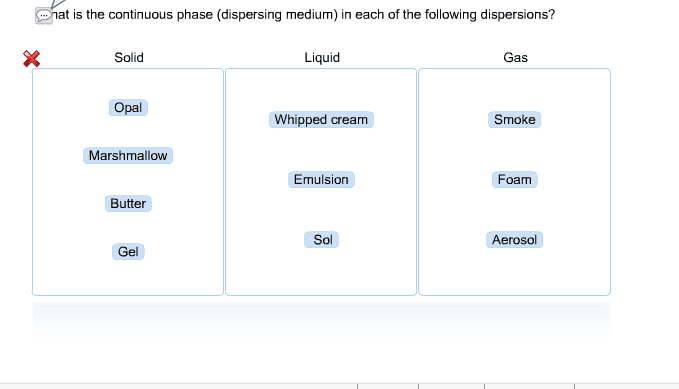
is the continuous phase (dispersing medium) in each of the following dispersions?

For unlimited access to Homework Help, a Homework+ subscription is required.
Related textbook solutions
Basic Chemistry
Principles of Chemistry Molecular Approach
Chemistry: Structure and Properties
Principles of Chemistry Molecular Approach
Chemistry: A Molecular Approach
Chemistry: A Molecular Approach
Principles of Chemistry: A Molecular Approach
Chemistry: The Central Science
Related questions
Define colloid.
| a mixture in which a substance is dissolved in a solvent | ||
| a mixture in which a substance settles to the bottom of a dispersing medium | ||
| a mixture in which a substance is dissolved in a solute | ||
| a mixture in which a dispersed substance is finely divided in a dispersing medium | ||
| a mixture in which a substance floats on the dispersing medium |
I need help with this question. Thankyou
Which of the following is NOT true.
| Colloid particles are generally larger than solute particles. | ||
| Over a long period of time colloid particles will settle out of the dispersing medium. | ||
| Colloids have dispersed particles that are large enough to scatter light. | ||
| A suspension have particles larger than colloid particles. |

