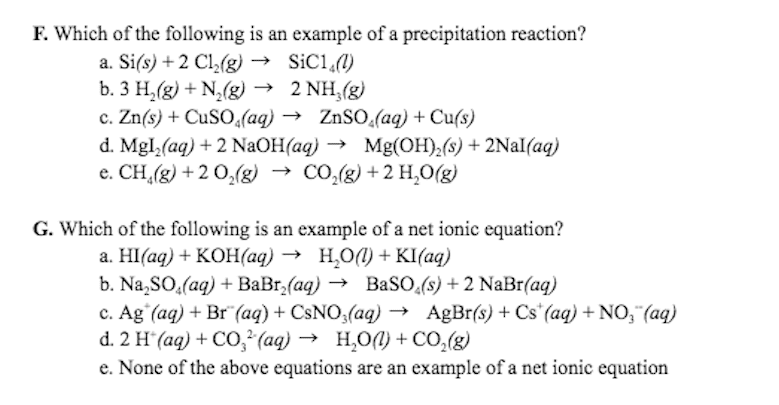A. Write a net ionic equation for all chemical reactions below.(Hint: it is possible for the net ionic equation to be the same asthe molecular equation. A complex ion that is in solution ismolecular in nature because units are coordinate covalently bondedtogether.)
1. Cu (s) + 4HNO3 (aq) --> Cu(NO3)2 (aq) + 2NO2 (g) + 2H2O(l)
2. Cu(NO3)2 (aq) + 2NaOH (aq) --> Cu(OH)2 (s) + 2NaNo3(aq)
3. Cu(OH)2 (s) --> CuO (s) + H2O (l)
4. CuO (s) + H2SO4 (aq) --> CuSO4 (aq) + H2O (l)
5. CuSO4 (aq) + Zn (s) --> Cu (s) + ZnSO4 (aq)
6. Zn (s) + H2SO4 (aq) --> ZnSO4 (aq) + H2 (g)
7. Zn (s) + 2HCl (aq) --> ZnCl2 (aq) + H2 (g)
B. Balance the following equations. Write each in both themolecular and net ionic form. Idenfity each reaction as anacid-base reaction, a precipitation reaction, anoxidation-reduction reaction, or as a combination of more than oneof these types.
1. ___Pb(NO3)2 (aq) + ____ NaCl (aq) --> _____PbCl2 (s) +_____NaNO3 (aq)
a) Molecular Equation:
b) Net Ionic Equation:
c) Reaction Type:
2. ___HNO3 (aq) + _____Zn(OH)2 (s) --> ____Zn(NO3)2 (aq) +____H2O (l)
a) Molecular Equation:
b) Net Ionic Equation:
c) Reaction Type:
THANK YOU!
A. Write a net ionic equation for all chemical reactions below.(Hint: it is possible for the net ionic equation to be the same asthe molecular equation. A complex ion that is in solution ismolecular in nature because units are coordinate covalently bondedtogether.)
1. Cu (s) + 4HNO3 (aq) --> Cu(NO3)2 (aq) + 2NO2 (g) + 2H2O(l)
2. Cu(NO3)2 (aq) + 2NaOH (aq) --> Cu(OH)2 (s) + 2NaNo3(aq)
3. Cu(OH)2 (s) --> CuO (s) + H2O (l)
4. CuO (s) + H2SO4 (aq) --> CuSO4 (aq) + H2O (l)
5. CuSO4 (aq) + Zn (s) --> Cu (s) + ZnSO4 (aq)
6. Zn (s) + H2SO4 (aq) --> ZnSO4 (aq) + H2 (g)
7. Zn (s) + 2HCl (aq) --> ZnCl2 (aq) + H2 (g)
B. Balance the following equations. Write each in both themolecular and net ionic form. Idenfity each reaction as anacid-base reaction, a precipitation reaction, anoxidation-reduction reaction, or as a combination of more than oneof these types.
1. ___Pb(NO3)2 (aq) + ____ NaCl (aq) --> _____PbCl2 (s) +_____NaNO3 (aq)
a) Molecular Equation:
b) Net Ionic Equation:
c) Reaction Type:
2. ___HNO3 (aq) + _____Zn(OH)2 (s) --> ____Zn(NO3)2 (aq) +____H2O (l)
a) Molecular Equation:
b) Net Ionic Equation:
c) Reaction Type:
THANK YOU!