2
answers
0
watching
338
views
28 Sep 2019
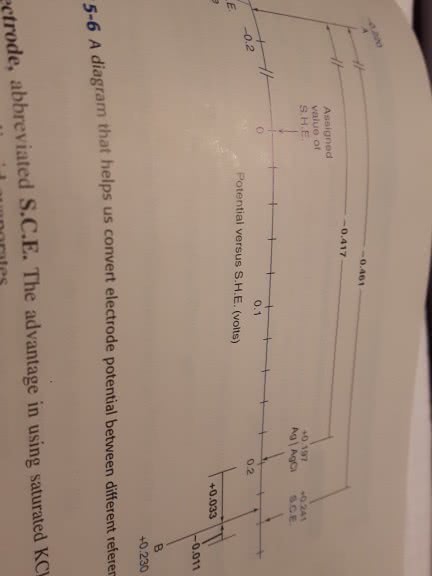
Convert the following voltages. Assume the silver-silverchloride and calomel reference electrodes are saturated with KCl,giving cell potentials of 0.197 V and 0.241, respectively.
(a) If an electrode has a potential of 0.659 V with respect to astandard hydrogen electrode (S.H.E.), what is the potential withrespect to a silver-silver chloride electrode?
(b) If an electrode has a potential of
Convert the following voltages. Assume the silver-silverchloride and calomel reference electrodes are saturated with KCl,giving cell potentials of 0.197 V and 0.241, respectively.
(a) If an electrode has a potential of 0.659 V with respect to astandard hydrogen electrode (S.H.E.), what is the potential withrespect to a silver-silver chloride electrode?
(b) If an electrode has a potential of
dcht24111997Lv10
16 Apr 2023
Keith LeannonLv2
28 Sep 2019
Already have an account? Log in