2
answers
0
watching
751
views
28 Sep 2019
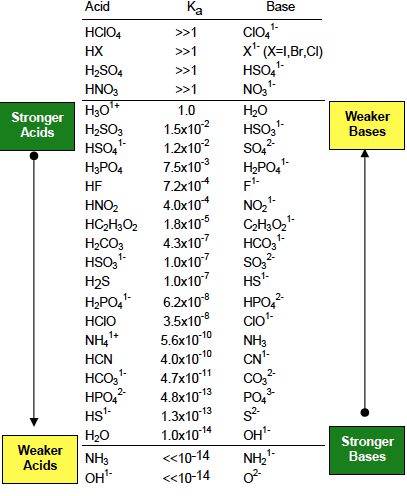
Write net Bronsted equations and determine the equilibriumconstants for the acid-base reactions that occur when aqueoussolutions of the following are mixed.
1. hydrazoic acid + sodium hypochlorite
2. KHSO4 + NH3
3. H2CO3 + KHS
Write net Bronsted equations and determine the equilibriumconstants for the acid-base reactions that occur when aqueoussolutions of the following are mixed.
1. hydrazoic acid + sodium hypochlorite
2. KHSO4 + NH3
3. H2CO3 + KHS
1. hydrazoic acid + sodium hypochlorite
2. KHSO4 + NH3
3. H2CO3 + KHS
Casey DurganLv2
28 Sep 2019
Already have an account? Log in