1
answer
1
watching
1,300
views
6 Nov 2019
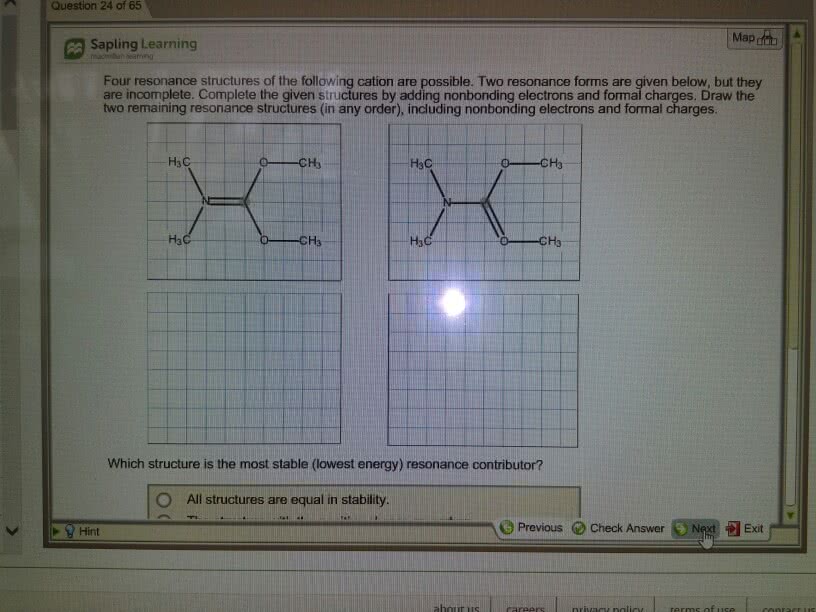
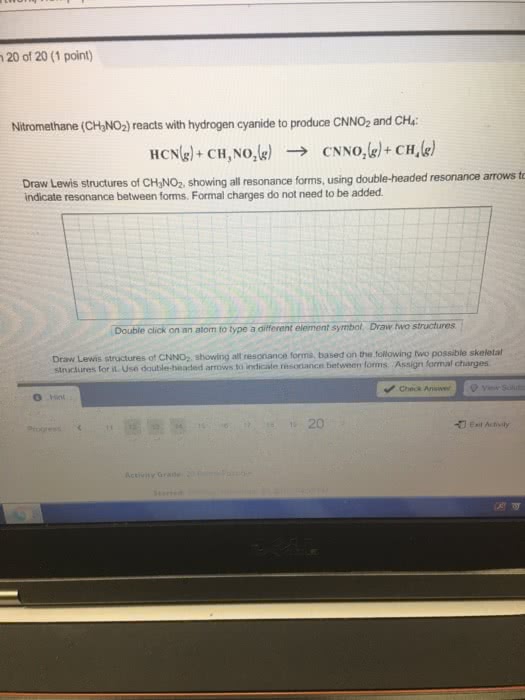
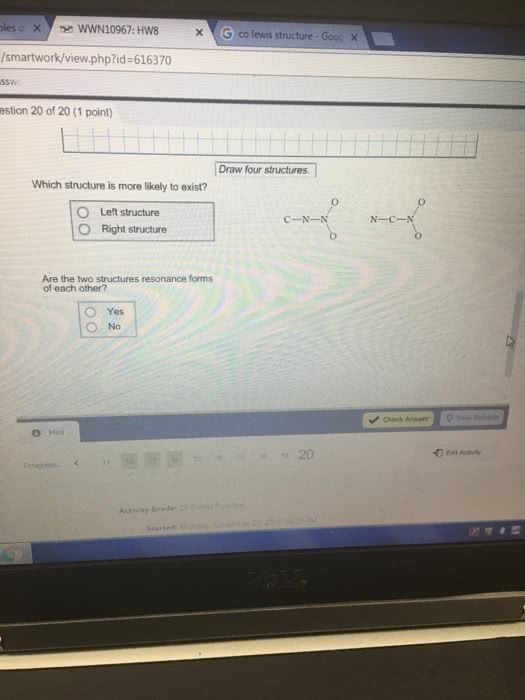
Draw the Kekule structures fir each of the following, showingall possible resonance forms with formal charges. Indicate thehybridization of all C and N atoms in each structures.
- (CH3)2CHCH3
- NO^+
- CH3CH2CO2^-
- CH3CHO
- C2H5OH
Draw the Kekule structures fir each of the following, showingall possible resonance forms with formal charges. Indicate thehybridization of all C and N atoms in each structures.
- (CH3)2CHCH3
- NO^+
- CH3CH2CO2^-
- CH3CHO
- C2H5OH
Nestor RutherfordLv2
10 Mar 2019