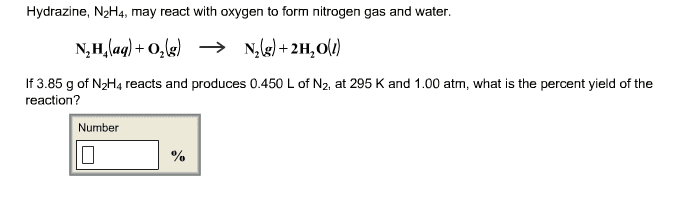1
answer
0
watching
547
views
6 Nov 2019
Hydrogen peroxide decomposes spontaneously to yield water andoxygen gas according to the following reaction equation.

The activation energy for this reaction is 75 kJ
2H2O2(aq) rightarrow 2H2O(l) + O2(g) Show transcribed image text
Hydrogen peroxide decomposes spontaneously to yield water andoxygen gas according to the following reaction equation.

The activation energy for this reaction is 75 kJ
2H2O2(aq) rightarrow 2H2O(l) + O2(g)
Show transcribed image text Tod ThielLv2
22 Mar 2019