1
answer
0
watching
515
views
6 Nov 2019
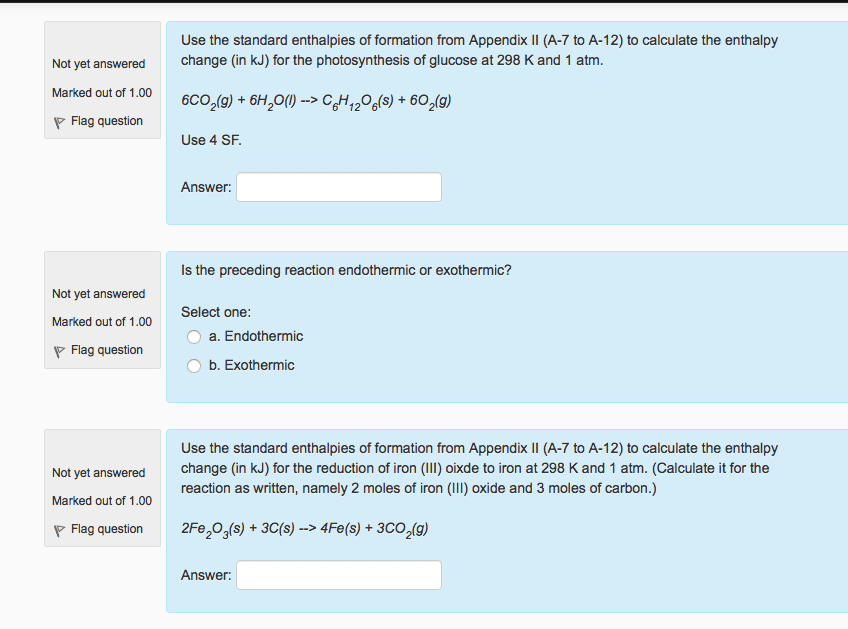
Use the standard enthalpies of formation from Appendix II (A-7 toA-12) to calculate the enthalpy change (in kJ) for the reduction ofiron (III) oixde to iron at 298 K and 1 atm. (Calculate it for thereaction as written, namely 2 moles of iron (III) oxide and 3 molesof carbon.)
2Fe2O3(s) + 3C(s) --> 4Fe(s) +3CO2(g)
Use the standard enthalpies of formation from Appendix II (A-7 toA-12) to calculate the enthalpy change (in kJ) for the reduction ofiron (III) oixde to iron at 298 K and 1 atm. (Calculate it for thereaction as written, namely 2 moles of iron (III) oxide and 3 molesof carbon.)
2Fe2O3(s) + 3C(s) --> 4Fe(s) +3CO2(g)
Elin HesselLv2
27 Jun 2019