2
answers
0
watching
241
views
greenmule713Lv1
6 Nov 2019
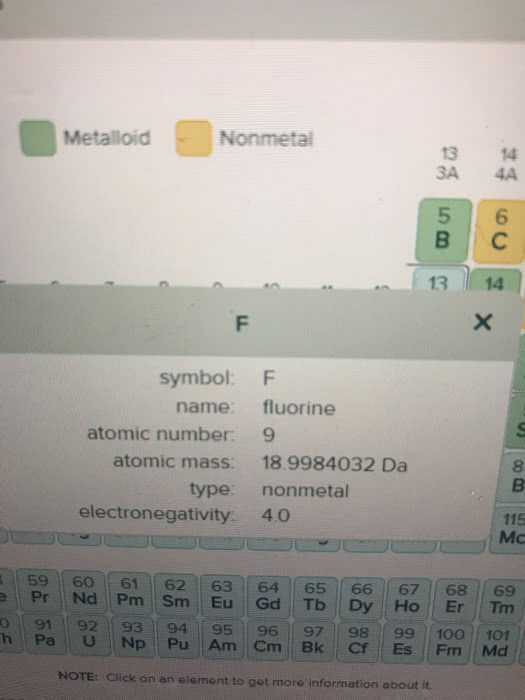
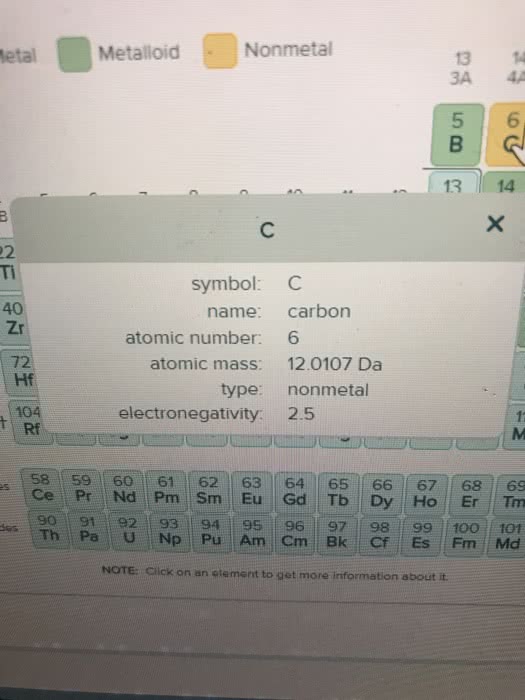
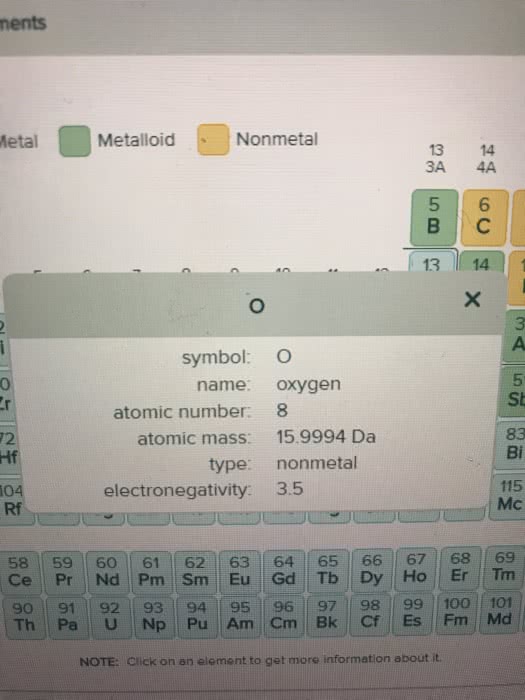
A 7.00L tank at 10.degree C is filled with 14.5 g of dinitrogenmonoxide gas and 3.41g of carbon dioxide gas. You can assume bothgases behave as ideal gases under these conditions.
Calculate the mole fraction of each gas. Be sure each of youranswer entries has the correct number of significant digits.
A 7.00L tank at 10.degree C is filled with 14.5 g of dinitrogenmonoxide gas and 3.41g of carbon dioxide gas. You can assume bothgases behave as ideal gases under these conditions.
Calculate the mole fraction of each gas. Be sure each of youranswer entries has the correct number of significant digits.
Calculate the mole fraction of each gas. Be sure each of youranswer entries has the correct number of significant digits.
Jamar FerryLv2
20 Apr 2019
Already have an account? Log in