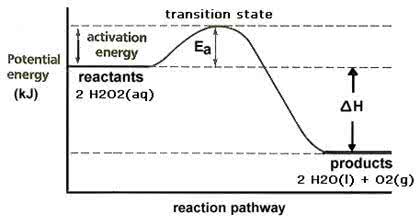
Hydrogen peroxide decomposes into water and oxygen by thefollowing reaction:
2 H2O2(aq) ? 2 H2O(l) + O2(g) ?Hrxn = -94.64 kJ/mol
Experimental evidence tells us that this reaction is first orderwith respect to hydrogen peroxide, with an activation energy of75.3 kJ/mol at 300 K.
(A.)What is the activation energy for the reverse reaction?
(B.)Sketch the reaction pathway below. Be sure to label the axes,reactants, products, transition state, and energies.
(C.)Assuming the forward and reverse reaction have the samefrequency factor, what is the equilibrium constant for thisreaction?
Catalase is an enzyme found in nearly all living organisms thatcatalyzes the hydrogen peroxide decomposition reaction shown aboveby lowering the activation energy of the forward reaction to 8.8kJ/mol.
(D.)How many times faster does hydrogen peroxide decompose inthe presence of catalase?
(E.)What temperature would we need to decompose hydrogen peroxodeat to achieve the same rate increase achieved with catalase?
Hydrogen peroxide decomposes into water and oxygen by thefollowing reaction:
2 H2O2(aq) ? 2 H2O(l) + O2(g) ?Hrxn = -94.64 kJ/mol
Experimental evidence tells us that this reaction is first orderwith respect to hydrogen peroxide, with an activation energy of75.3 kJ/mol at 300 K.
(A.)What is the activation energy for the reverse reaction?
(B.)Sketch the reaction pathway below. Be sure to label the axes,reactants, products, transition state, and energies.
(C.)Assuming the forward and reverse reaction have the samefrequency factor, what is the equilibrium constant for thisreaction?
Catalase is an enzyme found in nearly all living organisms thatcatalyzes the hydrogen peroxide decomposition reaction shown aboveby lowering the activation energy of the forward reaction to 8.8kJ/mol.
(D.)How many times faster does hydrogen peroxide decompose inthe presence of catalase?
(E.)What temperature would we need to decompose hydrogen peroxodeat to achieve the same rate increase achieved with catalase?