1
answer
0
watching
188
views
6 Nov 2019
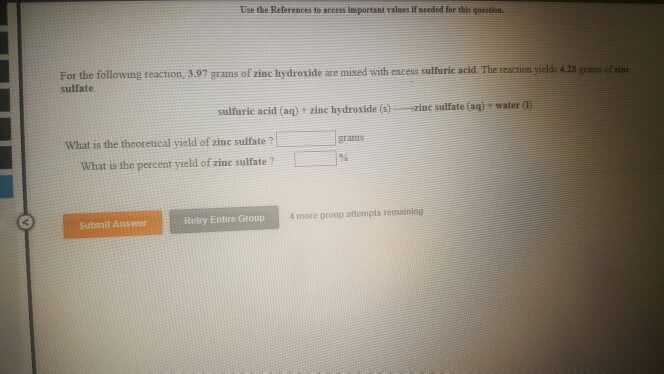
For the following reaction, 5.50 grams of water are mixed withexcess zinc oxide. The reaction yields 26.9 grams of zinchydroxide.
zinc oxide (s) + water (l) zinc hydroxide (aq)
What is the theoretical yield of zinc hydroxide ? grams
What is the percent yield for this reaction ?
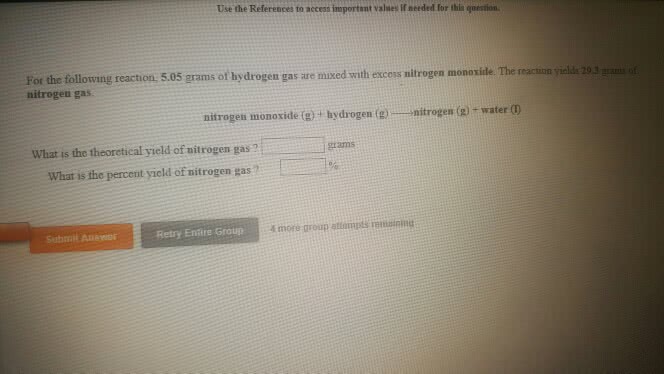
For the following reaction, 5.50 grams of water are mixed withexcess zinc oxide. The reaction yields 26.9 grams of zinchydroxide.
zinc oxide (s) + water (l) zinc hydroxide (aq)
What is the theoretical yield of zinc hydroxide ? grams
What is the percent yield for this reaction ?
Reid WolffLv2
2 May 2019