3
answers
0
watching
966
views
15 May 2018
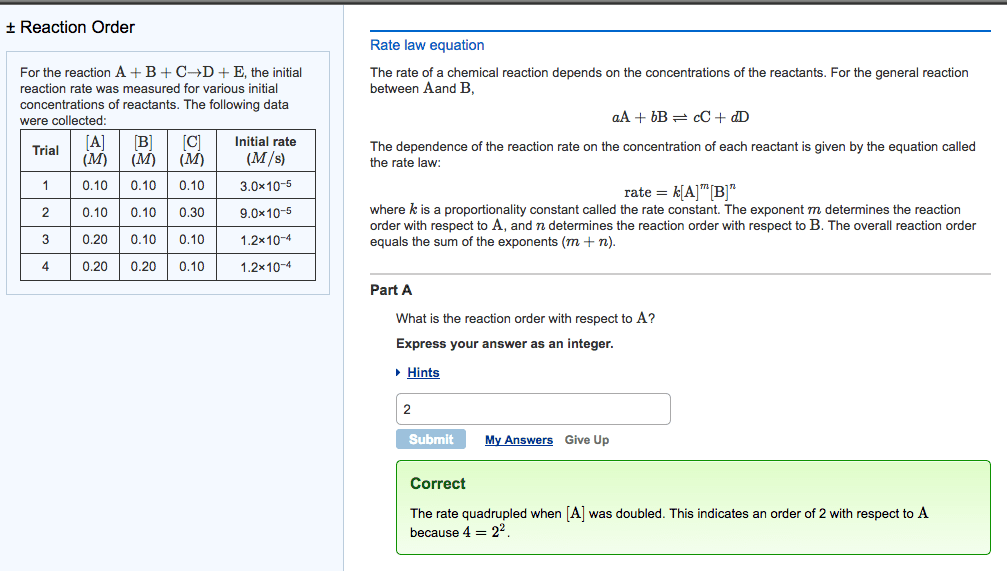
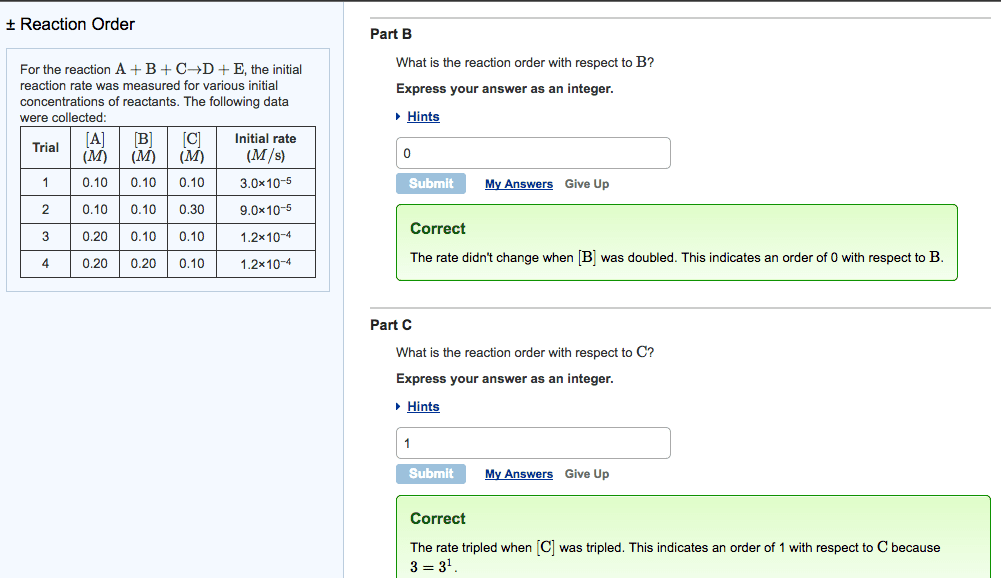
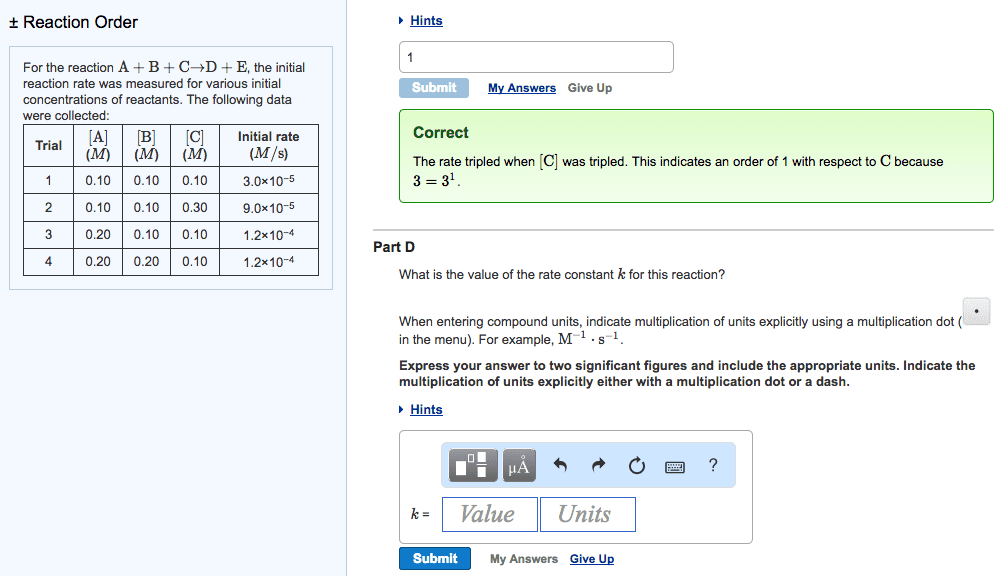
Q4. For the reaction 2 A + B - C, the initial rate is measured at several different reactant concentrations. From the resulting tabulated data, determine the rate law for the reaction. [A] (M) Initial Rate (M/s) 0.05 0.035 [B] (M) 0.05 0.05 0.10 0.10 0.070 0.20 0.56 a) Rate = k[A][B] c) Rate = k[A] [B] b) Rate = k[A] [B] d) Rate = k[A] [B]
Q4. For the reaction 2 A + B - C, the initial rate is measured at several different reactant concentrations. From the resulting tabulated data, determine the rate law for the reaction. [A] (M) Initial Rate (M/s) 0.05 0.035 [B] (M) 0.05 0.05 0.10 0.10 0.070 0.20 0.56 a) Rate = k[A][B] c) Rate = k[A] [B] b) Rate = k[A] [B] d) Rate = k[A] [B]
25 Jun 2023
Already have an account? Log in
Elin HesselLv2
15 May 2018
Already have an account? Log in