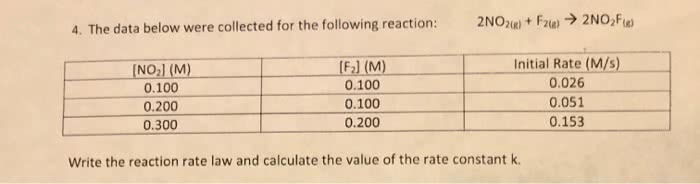2
answers
0
watching
415
views
18 May 2018
45. The tabulated data were collected for this reaction: 2 NO2(g) + F2(8) – 2 NO2F(8) [NO] [F] (M) Initial Rate (M/s) 0.100 0.100 0.026 0.200 0.100 0.051 0.200 0.200 0.103 0.400 0.400 0.411 Write an expression for the reaction rate law and calculate the value of the rate constant, k. What is the overall order of the reaction? On
45. The tabulated data were collected for this reaction: 2 NO2(g) + F2(8) – 2 NO2F(8) [NO] [F] (M) Initial Rate (M/s) 0.100 0.100 0.026 0.200 0.100 0.051 0.200 0.200 0.103 0.400 0.400 0.411 Write an expression for the reaction rate law and calculate the value of the rate constant, k. What is the overall order of the reaction? On
25 Jun 2023
Jean KeelingLv2
19 May 2018
Already have an account? Log in