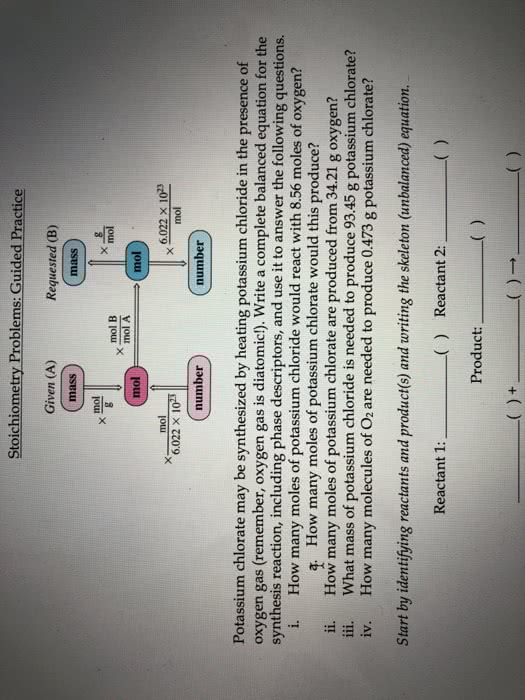
1
answer
0
watching
956
views
lemonyak871Lv1
28 Sep 2019
1.) how many mole of oxygen atoms are present in 5.36g of CaCo3?
2.) how many moles of Cl atoms are there in 1.7g of CCl4
3). What is the mass of 2.4x10^29 particles of CaCo3?
4.) In the reaction below, (CH4+2O2----> CO2 +2H2O) how many moles of CH4 are required to react with 0.5 mol of oxygen gas to form products?
5). Consider the reaction of 3 mol of P4 and 10 mol of oxygen gas to produce P2O5.
a. write the balance equation
b. how many moles of P2O5 could be produced from the reaction of 3 mol of P4 and 10 mol O2?
c.how many moles of excess reactant remain after the reaction is done?
1.) how many mole of oxygen atoms are present in 5.36g of CaCo3?
2.) how many moles of Cl atoms are there in 1.7g of CCl4
3). What is the mass of 2.4x10^29 particles of CaCo3?
4.) In the reaction below, (CH4+2O2----> CO2 +2H2O) how many moles of CH4 are required to react with 0.5 mol of oxygen gas to form products?
5). Consider the reaction of 3 mol of P4 and 10 mol of oxygen gas to produce P2O5.
a. write the balance equation
b. how many moles of P2O5 could be produced from the reaction of 3 mol of P4 and 10 mol O2?
c.how many moles of excess reactant remain after the reaction is done?
Nestor RutherfordLv2
28 Sep 2019