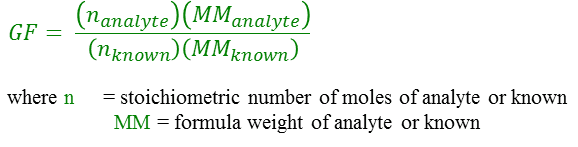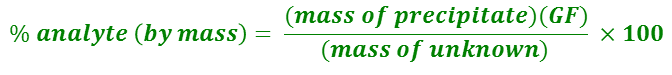1
answer
0
watching
548
views
copperfox95Lv1
28 Sep 2019
I'm doing a chemistry lab on gravimetric analysis of a group 1 metal carbonate compound... So basically, we heated the unknown metal carbonate, and then mixed it with calcium chloride solution. Then, we dried and weighed these products and recorded their masses. I'm having trouble finding the final mass & identity of the precipitate, alongside its molar mass.
My balanced chemical equation is: CaCl2 + X2CO3 (where X = unknown group 1 metal) --> CaCO3 + 2XCl
The mass of the heated compound was 2.054-1.001 (where 1.001 = mass of filter paper) in the beginning and 2.242-1.001 at the end. The mass of the non-heated compound was 2.008 in the beginning and 2.325 in the end.
Thanks in advance!
I'm doing a chemistry lab on gravimetric analysis of a group 1 metal carbonate compound... So basically, we heated the unknown metal carbonate, and then mixed it with calcium chloride solution. Then, we dried and weighed these products and recorded their masses. I'm having trouble finding the final mass & identity of the precipitate, alongside its molar mass.
My balanced chemical equation is: CaCl2 + X2CO3 (where X = unknown group 1 metal) --> CaCO3 + 2XCl
The mass of the heated compound was 2.054-1.001 (where 1.001 = mass of filter paper) in the beginning and 2.242-1.001 at the end. The mass of the non-heated compound was 2.008 in the beginning and 2.325 in the end.
Thanks in advance!
Sixta KovacekLv2
28 Sep 2019