4
answers
0
watching
2,258
views
25 Aug 2018
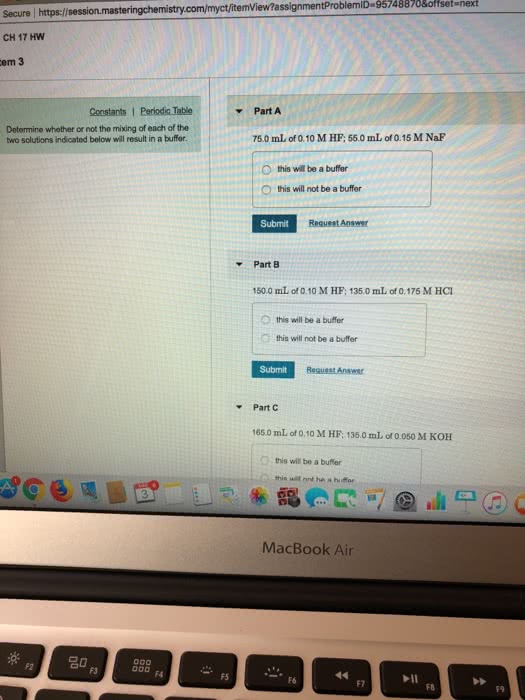
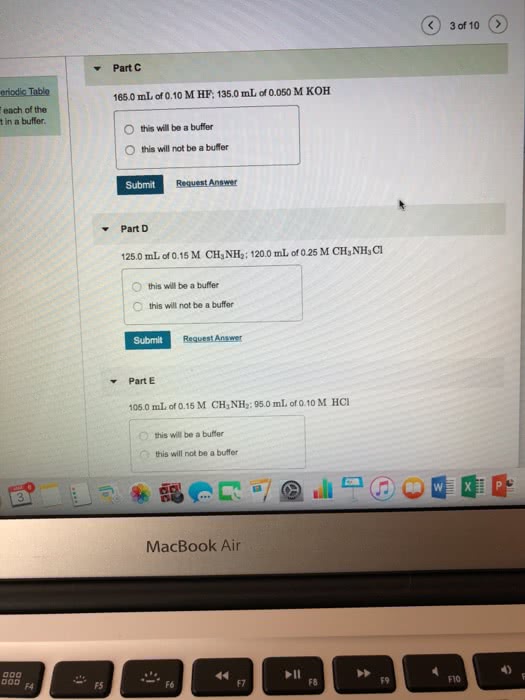
53. Determine whether or not the mixing of each pair of solutions results in a buffer. a. 100.0 mL of 0.10 M NH3; 100.0 mL of 0.15 M NH CI b. 50.0 mL of 0.10 M HCl; 35.0 mL of 0.150 M NaOH c. 50.0 mL of 0.15 M HF; 20.0 mL of 0.15 M NaOH d. 175.0 mL of 0.10 M NH3; 150.0 mL of 0.12 M NaOH e. 125.0 mL of 0.15 M NH3; 150.0 mL of 0.20 M NaOH
53. Determine whether or not the mixing of each pair of solutions results in a buffer. a. 100.0 mL of 0.10 M NH3; 100.0 mL of 0.15 M NH CI b. 50.0 mL of 0.10 M HCl; 35.0 mL of 0.150 M NaOH c. 50.0 mL of 0.15 M HF; 20.0 mL of 0.15 M NaOH d. 175.0 mL of 0.10 M NH3; 150.0 mL of 0.12 M NaOH e. 125.0 mL of 0.15 M NH3; 150.0 mL of 0.20 M NaOH
Liked by bayanmabuhamdia
Keith LeannonLv2
26 Aug 2018
Read by 2 people
hardiksahuLv10
17 May 2023
Already have an account? Log in
Read by 2 people
Read by 2 people