1
answer
0
watching
2,052
views
5 Nov 2019

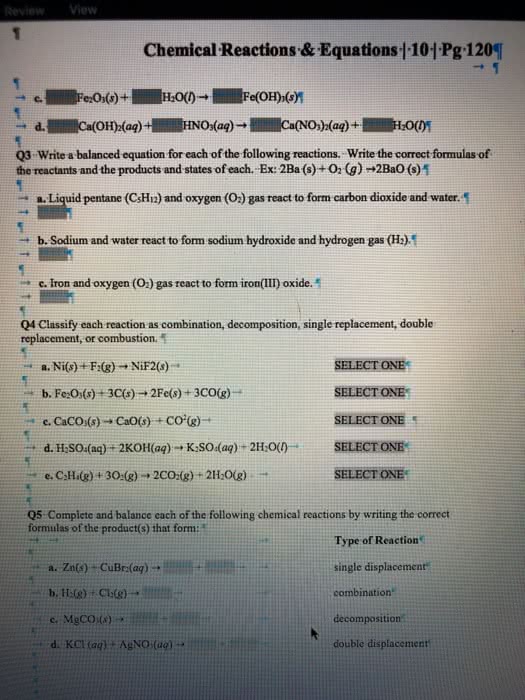
Classify each chemical reaction: type of reaction (check all that apply) reaction combination l precipitation single replacement combustion 2H20(g) â 2H2(g) + O2(g) O double replacement acid-base decomposition combination single replacement O precipitation combustion Mg12(ag) + Pb(NO3)2(aq) â Mg(NO3)2(ag) + PbI-(s) O double replacement acid-base decomposition combination single replacement double replacement decomposition precipitation combustion acid-base KOH (aq)-HClO4(aq) â KCIO4(aq) + H2O(I) lprecipitation combustion acid-base Ucombination O single replacement 2C113(CH2)2CH3(g) + 1302(g) â 8CO2(g) + 101120(g) double replacement decomposition

Classify each chemical reaction: type of reaction (check all that apply) reaction combination l precipitation single replacement combustion 2H20(g) â 2H2(g) + O2(g) O double replacement acid-base decomposition combination single replacement O precipitation combustion Mg12(ag) + Pb(NO3)2(aq) â Mg(NO3)2(ag) + PbI-(s) O double replacement acid-base decomposition combination single replacement double replacement decomposition precipitation combustion acid-base KOH (aq)-HClO4(aq) â KCIO4(aq) + H2O(I) lprecipitation combustion acid-base Ucombination O single replacement 2C113(CH2)2CH3(g) + 1302(g) â 8CO2(g) + 101120(g) double replacement decomposition
Collen VonLv2
21 Apr 2020