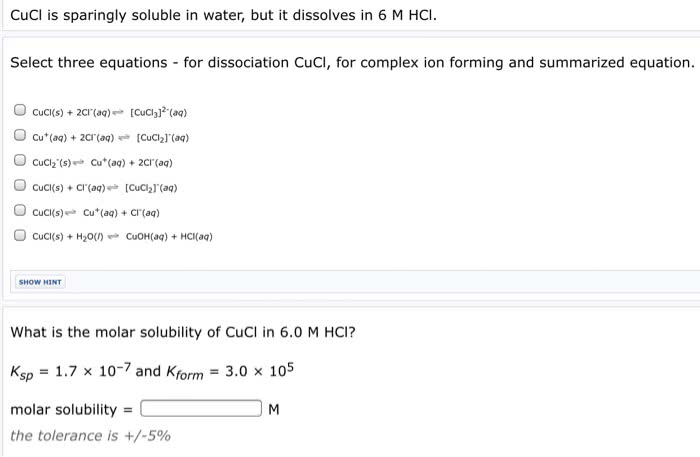
3
answers
0
watching
820
views
10 Nov 2017
94. The solubility of copper(I) chloride is 3.91 mg per 100.0 mL of solution. Calculate Ksp for CuCl.
94. The solubility of copper(I) chloride is 3.91 mg per 100.0 mL of solution. Calculate Ksp for CuCl.
13 Jun 2023
Joey Tang
Bachelor’s Degree in Chemistry from McMaster University10 Jun 2020
Answer verification
This is a step by step verification of the answer by our certified expert.
Subscribe to our livestream channel for more helpful videos.