3
answers
0
watching
1,920
views
24 Mar 2018
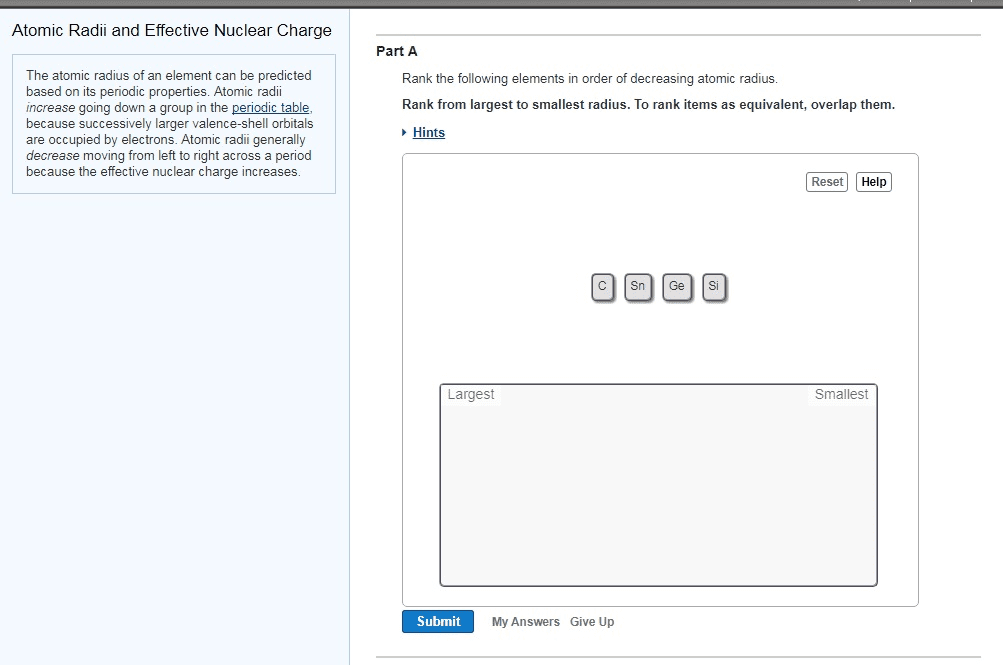
137. The atomic radii of the isotopes of an element are identical to one another. However, the atomic radii of the ions of an ele- ment are significantly different from the atomic radii of the neutral atom of the element. Explain.
137. The atomic radii of the isotopes of an element are identical to one another. However, the atomic radii of the ions of an ele- ment are significantly different from the atomic radii of the neutral atom of the element. Explain.
24 Jun 2023
Elin HesselLv2
24 Mar 2018
Already have an account? Log in