1
answer
2
watching
1,001
views
orchidelk884Lv1
28 Sep 2019
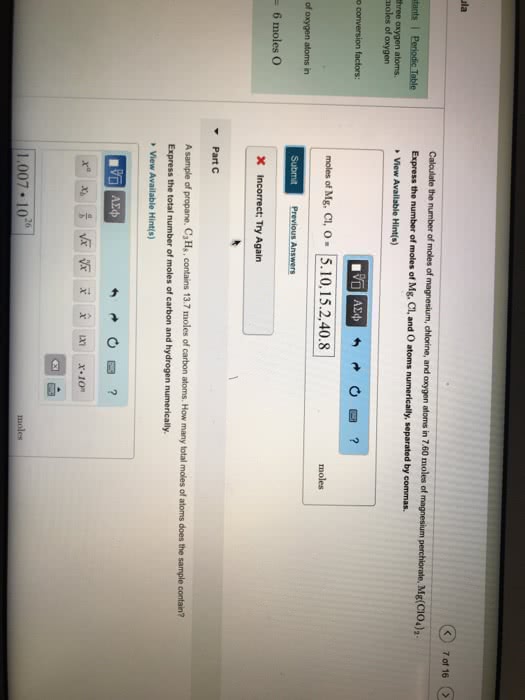
Calculate the number of moles of magnesium, chlorine, and oxygen atoms in 7.90 moles of magnesium perchlorate, Mg(ClO4)2. Express the number of moles of Mg, Cl, and O atoms numerically, separated by commas.
Calculate the number of moles of magnesium, chlorine, and oxygen atoms in 7.90 moles of magnesium perchlorate, Mg(ClO4)2. Express the number of moles of Mg, Cl, and O atoms numerically, separated by commas.
Liked by anayanci.hernandez
Collen VonLv2
28 Sep 2019