1
answer
0
watching
618
views
28 Sep 2019
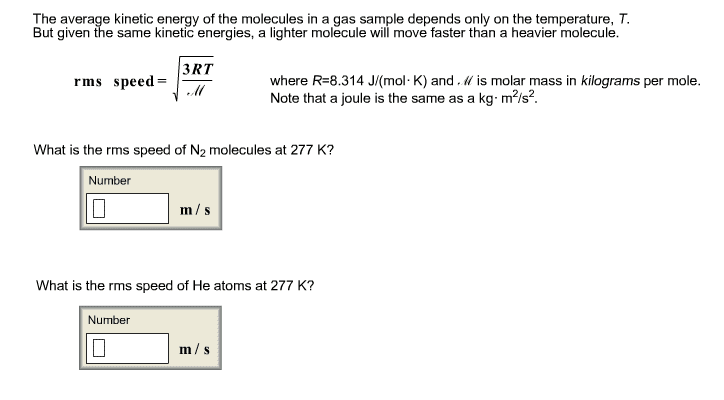
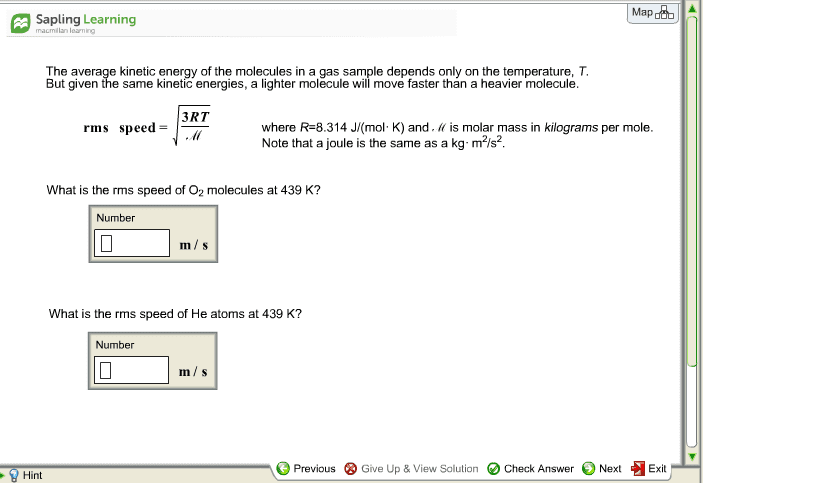
The average kinetic energy of the molecules in a gas sample depends only on the temperature, T. But given the same kinetic energies, a lighter molecule will move faster than a heavier molecule. where R=8.314 J/(mol·K) and Ⳡis molar mass in kilograms per mole. Note that a joule is the same as a kg·m2/s2. 1.What is the rms speed of F2 molecules at 465 K? 2.What is the rms speed of He atoms at 465 K?
The average kinetic energy of the molecules in a gas sample depends only on the temperature, T. But given the same kinetic energies, a lighter molecule will move faster than a heavier molecule. where R=8.314 J/(mol·K) and Ⳡis molar mass in kilograms per mole. Note that a joule is the same as a kg·m2/s2. 1.What is the rms speed of F2 molecules at 465 K? 2.What is the rms speed of He atoms at 465 K?
Sixta KovacekLv2
30 Sep 2019