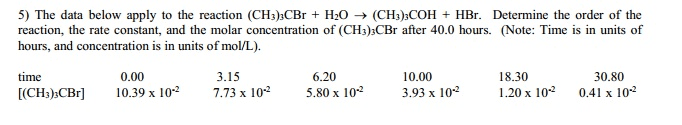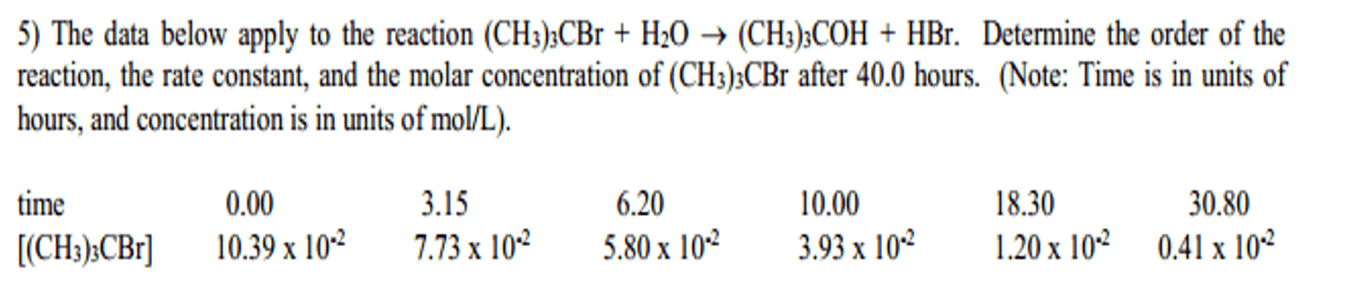2
answers
1
watching
1,638
views
28 Sep 2019
The reaction of tert-butyl-bromide (CH3)3CBr with water is represented by the equation:
(CH3)3CBr + H2O â (CH3)3COH + HBr
The following data were obtained from three experiments using the method of initial rates:
Initial [(CH3)3CBr] mol L-1 Initial [H2O] mol L-1 Initial rate mol L-1 min-1 Experiment 1 5.0 x 10^-2 2.0 x 10^-2 2.0 x 10-6 Experiment 2 5.0 x 10-2 4.0 x 10-2 2.0 x 10-6 Experiment 3 1.0 x 10-1 4.0 x 10-2 4.0 x 10-6
a. What is the order with respect to (CH3)3CBr?
b. What is the order with respect to H2O?
c. What is the overall order of the reaction?
d. Write the rate equation.
e. Calculate the rate constant k for the reaction
The reaction of tert-butyl-bromide (CH3)3CBr with water is represented by the equation:
(CH3)3CBr + H2O â (CH3)3COH + HBr
The following data were obtained from three experiments using the method of initial rates:
| Initial [(CH3)3CBr] mol L-1 | Initial [H2O] mol L-1 | Initial rate mol L-1 min-1 | |
| Experiment 1 | 5.0 x 10^-2 | 2.0 x 10^-2 | 2.0 x 10-6 |
| Experiment 2 | 5.0 x 10-2 | 4.0 x 10-2 | 2.0 x 10-6 |
| Experiment 3 | 1.0 x 10-1 | 4.0 x 10-2 | 4.0 x 10-6 |
a. What is the order with respect to (CH3)3CBr?
b. What is the order with respect to H2O?
c. What is the overall order of the reaction?
d. Write the rate equation.
e. Calculate the rate constant k for the reaction
Liked by esmaelziyad946 and 2 others
Jean KeelingLv2
29 Sep 2019
Already have an account? Log in