CHEM 1211 Lecture Notes - Lecture 4: Gibbs Free Energy, Calorimetry, Thermochemistry
Document Summary
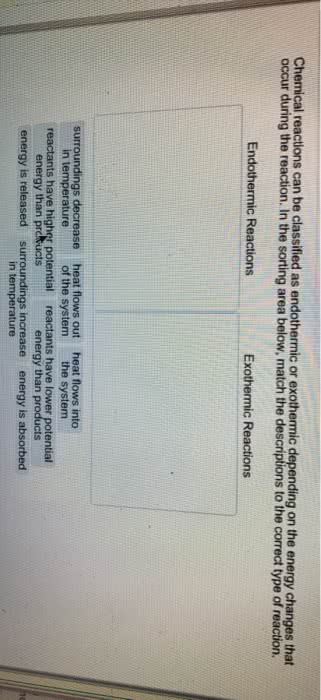
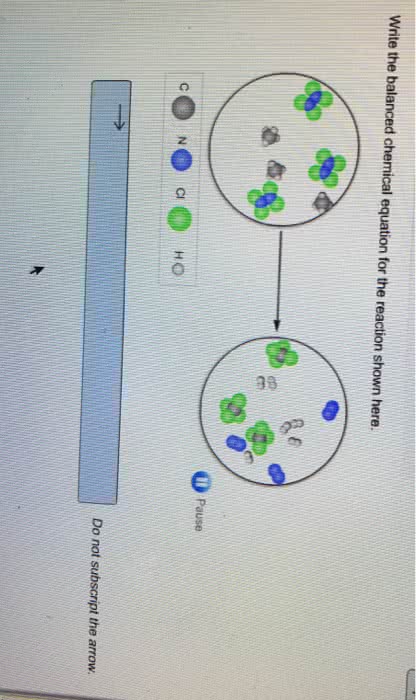
Chem 1211 chapter 6: **energy**: energy is the capacity to do work or transfer heat. Energy can exist in various forms, such as kinetic energy (motion) and potential energy (position or composition). The si unit of energy is the joule (j): **first law of thermodynamics**: this law states that energy cannot be created or destroyed, only converted from one form to another. Energy can be exchanged between the system and the surroundings as heat or work: **heat (q)**: heat is the transfer of energy between a system and its surroundings due to a temperature difference. In chemistry, work is often related to the expansion or compression of gases: **enthalpy (h)**: enthalpy is a state function that represents the heat content of a system at constant pressure. Enthalpy changes ( h) are used to describe the heat absorbed or released during chemical reactions: **calorimetry**: calorimetry is an experimental technique used to measure the heat involved in a chemical or physical process.