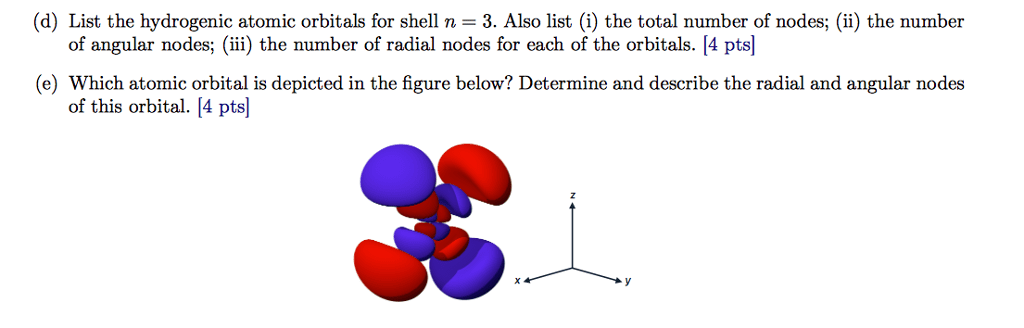CHEM 101 Lecture Notes - Bohr Radius, Polynomial, Uncertainty Principle
Document Summary
Y tells whereits likelyto be ne at ro but this can"t happen. 14 n size of orbital l shape me direction meme. Radialor angularor shape s orbital p orbital d orbital. 2 l nodes nodes n 1 angular nodes l g of radial nodes n l. 1 2 angular nodes are the same no matterif 10p or sp l. Il if l o then me can only be 0. There are n wavefunctions h 2 has 4 wavefunctions a wavefunctions h 3 has. Degeneracy lowerenergy more stable ne z t d d e 0 then based on l youcan find me values. Energy of electronic states is discrete but position is continuous. Electron doesn"t fall into nucleus because increased certainty ke a violate the uncertainty principle. As n increases energy in orbital increases orbital gets larger. Rdf peak is the bohr radius my itr gfurface area of sphere certaindistance is the peak of this graph radial most probable shell.