CHEM-1601 Chapter Notes - Chapter 5: Bond Dipole Moment, Chemical Polarity, Ionic Bonding
Document Summary
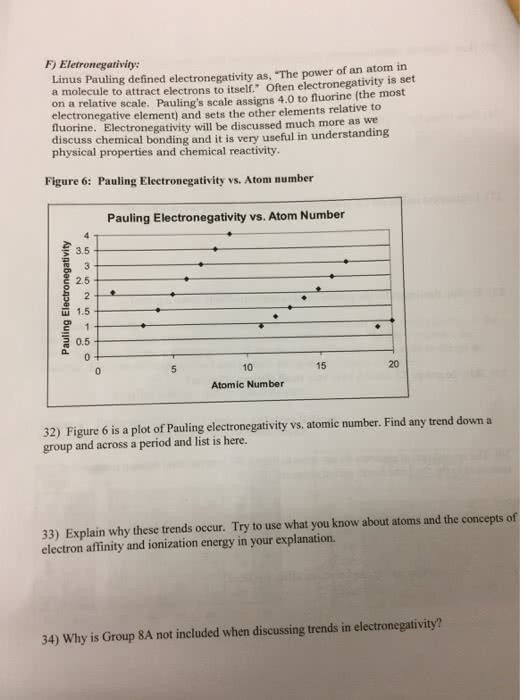
What are the periodic trends in electronegativity: electronegativity: the ability of an atom to attract electrons to itself in a chemical bond, electronegativities are positive values, periodic trends: i. ii. Decreases down a column: f is most electronegative and fr is least electronegative, electronegativities to memorize: i. F = 4. 0: o = 3. 5 iii. iv, h = 2. 1. C = s = 2. 5: explain the difference between a pure covalent bond, a polar covalent bond, and an ionic bond, degree of polarity in a chemical bond depends on electronegativity difference between the bonding atoms i. The greater the difference, the more polar the bond: pure (nonpolar) covalent bond: i. ii. iii. Ex: cl2: polar covalent bond: i. ii. Electrons shared unequally (has a positive pole and a negative pole) Ex: use dipole moment formula to calculate that separating a p+ and e- by 130 pm results in dipole moment = 6. 2 d.